Chapter 41: Q. 23 (page 1207)
A helium-neon laser emits a visible laser beam with a wavelength of . How many photons are emitted per second?
Short Answer
No of photons emitted per second are
Learning Materials
Features
Discover
Chapter 41: Q. 23 (page 1207)
A helium-neon laser emits a visible laser beam with a wavelength of . How many photons are emitted per second?
No of photons emitted per second are
All the tools & learning materials you need for study success - in one app.
Get started for free
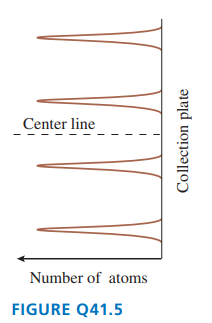
FIGURE Qshows the outcome of a Stern-Gerlach experiment with atoms of element .
a. Do the peaks represent different values of the atom’s total angular momentum or different values of the component of its angular momentum? Explain.
b. What angular momentum quantum numbers characterize these four peaks?
A neon discharge tube emits a bright reddish-orange spectrum, but a glass tube filled with neon is completely transparent. Why doesn’t the neon in the tube absorb orange and red wavelengths?
What is an atom’s ionization energy? In other words, if you know the ionization energy of an atom, what is it that you know about the atom?
An atom in an excited state has a chance of emitting a photon in . How long will it take for of a sample of excited atoms to decay?
The hydrogen atomwave function is a maximum at. But the radial probability density, shown peaks at and is zero at. Explain this paradox.
What do you think about this solution?
We value your feedback to improve our textbook solutions.