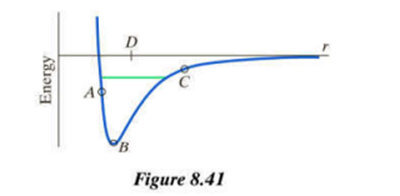
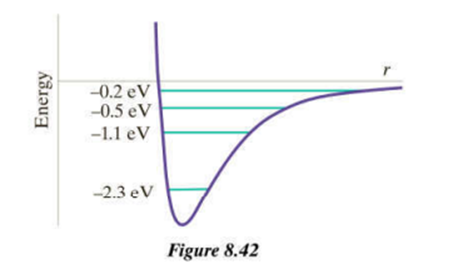
Chapter 8: Q26P (page 345)
Make a rough estimate of this uniform energy spacing in electron volts (where ). You will need to make some rough estimates of atomic properties based on prior work. For comparison with the spacing of these vibrational energy states, note that the spacing between quantized energy levels for "electronic" states such as in atomic hydrogen is of the order of several electron volts.
(b) List several photon energies that would be emitted if a number of these vibrational energy levels were occupied due to collisional excitation. To what region of the spectrum (x-ray, visible, microwave, etc.) do these photons belong? (See Figure 8.1 at the beginning of the chapter.)

Short Answer
a. The uniform energy spacing is .
b. The several photon energies are , and respectively. The photons belong in the spectrum’s infrared part.