Chapter 8: Q25P (page 346)
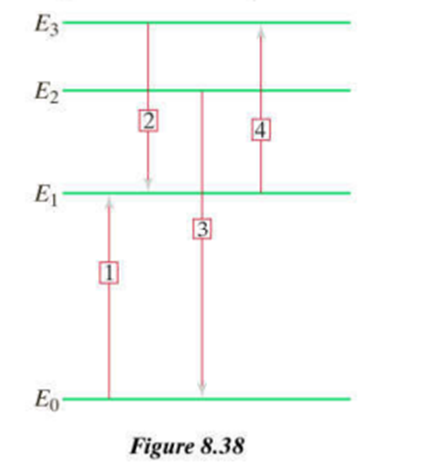
Consider a microscopic spring–mass system whose spring stiffness is, and the mass is. (a) What is the smallest amount of vibrational energy that can be added to this system? (b) What is the difference in mass (if any) of the microscopic oscillator between being in the ground state and being in the first excited state? (c) In a collection of these microscopic oscillators, the temperature is high enough that the ground state and the first three excited states are occupied. What are possible energies of photons emitted by these oscillators?