Chapter 18: 47P (page 542)
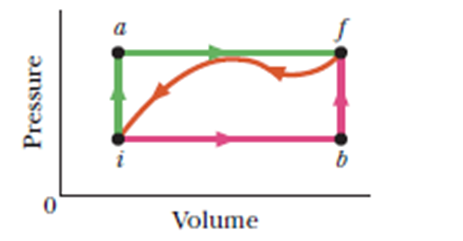
When a system is taken from state i to state f along path iaf in figure, Q=50 cal and W=20 cal. Along path ibf, Q=36 cal. (a) What is W along path ibf? (b) If W= - 13 calfor the return path fi, what is Q for this path? (c) If Eint,i=10 cal, what is Eint,f? If Eint,b=22 cal, (d) what is Q for path ib and (e) what is Q for path bf?
Short Answer
- The work done along path ibf is 6 cal
- The heat produced for path fi is -43 cal
- The value of is
- The value of heat for path ib is 18 cal
- The heat produced for path bf is 18 cal