Chapter 18: Q73P (page 546)
What is the volume increase of an aluminum cubeon an edge when heated fromto?
Short Answer
The volume increase of an aluminum cube is
Learning Materials
Features
Discover
Chapter 18: Q73P (page 546)
What is the volume increase of an aluminum cubeon an edge when heated fromto?
The volume increase of an aluminum cube is
All the tools & learning materials you need for study success - in one app.
Get started for free
An energetic athlete can use up all the energy from a diet of . If he were to use up this energy at a steady rate, what is the ratio of the rate of energy use compared to that of abulb? (The power ofis the rate at which the bulb converts electrical energy to heat and the energy of visible light.)
A small electric immersion heater is used to heat of water for a cup of instant coffee. The heater is labeled “” (it converts electrical energy to thermal energy at this rate). Calculate the time required to bring all this water from, ignoring any heat losses.
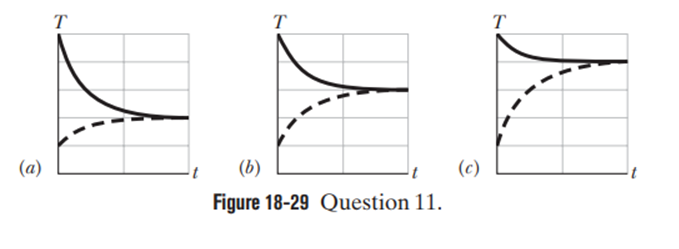
A hot object is dropped into a thermally insulated container of water, and the object and water are then allowed to come to thermal equilibrium. The experiment is repeated twice, with different hot objects. All three objects have the same mass and initial temperature and the mass and initial temperature of the water are the same in the three experiments. For each of the experiments, Fig. 18-29 gives graphs of the temperatures Tof the object and the water versus time t. Rankthe graphs according to the specific heats of the objects, greatest first.
Calculate the specific heat of a metal from the following data. A container made of the metal has a mass of and containsof water. Apiece of the metal initially at a temperature ofis dropped into the water. The container and water initially have a temperature of, and the final temperature of the entire (insulated) system is.
What mass of steam at must be mixed with of ice at its melting point, in a thermally insulated container, to produce liquid water at ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.