Chapter 18: Q31P (page 542)
What mass of steam at must be mixed with of ice at its melting point, in a thermally insulated container, to produce liquid water at ?
Short Answer
The mass of the steam is
Learning Materials
Features
Discover
Chapter 18: Q31P (page 542)
What mass of steam at must be mixed with of ice at its melting point, in a thermally insulated container, to produce liquid water at ?
The mass of the steam is
All the tools & learning materials you need for study success - in one app.
Get started for free
The Pyrex glass mirror in a telescope has a diameter of 170 in.The temperature ranges fromtoon the location of the telescope. What is the maximum change in the diameter of the mirror, assuming that the glass can freely expand and contract?
One way to keep the contents of a garage from becoming too cold on a night when a severe subfreezing temperature is forecast is to put a tub of water in the garage. If the mass of the water is and its initial temperature is, (a) how much energy must the water transfer to its surroundings in order to freeze completely and (b) what is the lowest possible temperature of the water and its surroundings until that happens?
Question: A0.300 kg sample is placed in a cooling apparatus that removes energy as heat at a constant rate of 2.81 W . Figure 18-52 gives the temperature Tof the sample versus time t.The temperature scale is set by and the time scale is set by .What is the specific heat of the sample?
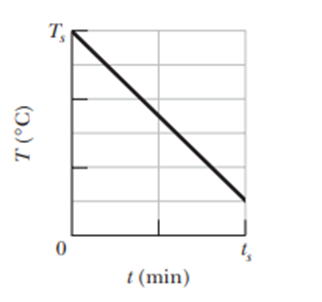
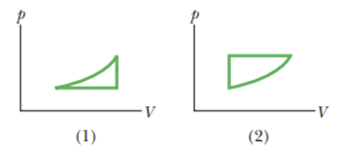
Figure 18-27 shows two closed cycles on p-Vdiagrams for a gas. The three parts of cycle 1 are of the same length and shape as those of cycle 2. For each cycle, should the cycle be traversed clockwise or counterclockwise if (a) the net work done by the gas is to be positive and (b) the net energy transferred by the gas as heatis to be positive?
An energetic athlete can use up all the energy from a diet of . If he were to use up this energy at a steady rate, what is the ratio of the rate of energy use compared to that of abulb? (The power ofis the rate at which the bulb converts electrical energy to heat and the energy of visible light.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.