Chapter 18: Q74P (page 546)
In a series of experiments, block Bis to be placed in a thermally insulated container with block A, which has the same mass as block B. In each experiment, block Bis initially at a certain temperature, but temperature of block Ais changed from experiment to experiment. Let represent the final temperature of the two blocks when they reach thermal equilibrium in any of the experiments.
Figure 18-53 gives temperature versus the initial temperature for a range of possible values of , from to. The vertical axis scale is set by . What are (a) temperature and (b) the ratio of the specific heats of the blocks?
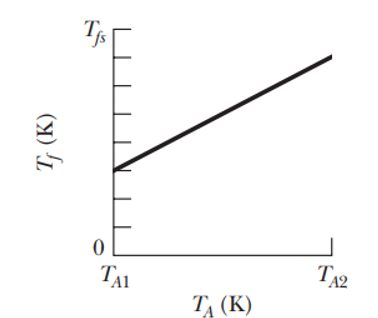
Figure 18.53
Short Answer
- The temperature of block B is.
- The ratio of the specific heats of the blocks is 1.5