Chapter 18: 16P (page 541)
When the temperature of a metal cylinder is raised from, its length increases by 0.23%
(a) Find the percent change in density.
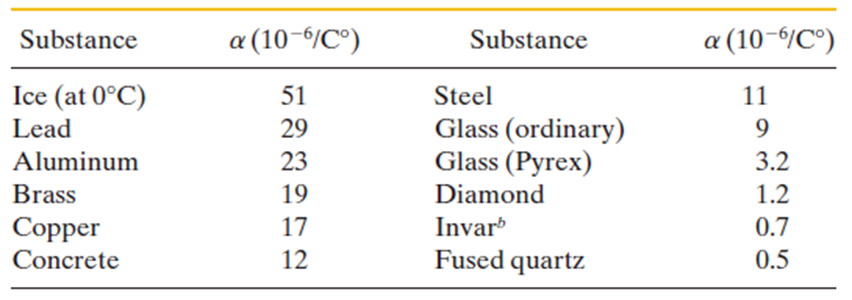
(b) What is the metal? Use Table.
Short Answer
- The percent change in density is -0.69%
- The coefficient of expansion is . The given metal is Aluminum.