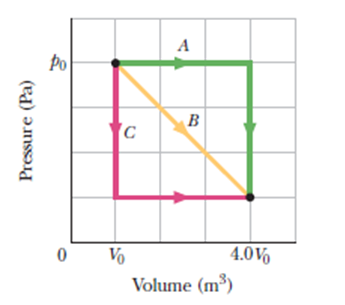
Chapter 18: Q69P (page 545)
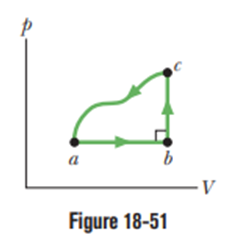
Figure 18-51 displays a closed cycle for a gas. The change in internal energy along pathis . The energy transferred to the gas as heat is along path ab, and along path . How much work is done by the gas along (a) path abcand (b) path ab?
Short Answer
- The work done by the gas along path abc is
- The work done by the gas along path ab is