Chapter 18: 50P (page 542)
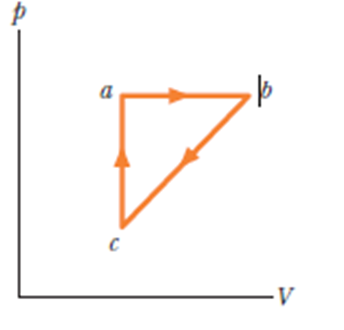
A lab sample of gas is taken through cycle abca shown in the p-V diagram of Figure. The net work done is +1.2 J. Along path ab, the change in the internal energy is +3.0 Jand the magnitude of the work done is 5.0 J. Along path ca, the energy transferred to the gas as heat is +2.5 J.
- How much energy is transferred as heat along path ab and
- How much energy is transferred as heat along path bc?
Short Answer
- Energy transferred as heat along path ab is 8.0 J
- Energy transferred as heat along path bc is -9.3 J