Chapter 18: Q91P (page 547)
It is possible to melt ice by rubbing one block of it against another. How much work, in joules, would you have to do to getof ice to melt?
Short Answer
The work to be done to melt of ice is.
Learning Materials
Features
Discover
Chapter 18: Q91P (page 547)
It is possible to melt ice by rubbing one block of it against another. How much work, in joules, would you have to do to getof ice to melt?
The work to be done to melt of ice is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Soon after Earth was formed, heat released by the decay of radioactive elements raised the average internal temperature from, at about which value it remains today. Assuming an average coefficient of volume expansion of, by how much has the radius of Earth increased since the planet was formed?
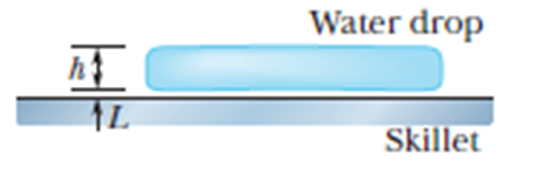
Leidenfrost Effect. A water drop that is slung onto a skillet with a temperature between and about will last about . However, if the skillet is much hotter, the drop can last several minutes, an effect named after an early investigator. The longer lifetime is due to the support of a thin layer of air and water vapor that separates the drop from the metal (by distance L in Figure). Let , and assume that the drop is flat withheight and bottom face area . Also assume that the skillet has a constant temperature and the drop has a temperature of .Water has density , and the supporting layer has thermal conductivity . (a) At what rate is energy conducted from the skillet to the drop through the drop’s bottom surface? (b) If conduction is the primary way energy moves from the skillet to the drop, how long will the drop last?
A thermometer of mass and of specific heat reads . It is then completely immersed in of water, and it comes to the same final temperature as the water. If the thermometer then reads , what was the temperature of the water before insertion of the thermometer?
Penguin huddling. To withstand the harsh weather of the Antarctic, emperor penguins huddle in groups (Figure). Assume that a penguin is a circular cylinder with a top surface area and height . Let be the rate at which an individual penguin radiates energy to the environment (through the top and the sides); thus is the rate at which N identical, well-separated penguins radiate. If the penguins huddle closely to form a huddled cylinder with top surface area and height , the cylinder radiates at the rate . If , (a) what is the value of the fraction and (b) by what percentage does huddling reduce the total radiation loss?

At , a rod is exactlylong on a steel ruler. Both the rod and the ruler are placed in an oven at, where the rod now measureson the same ruler. What is the coefficient of linear expansion for the material of which the rod is made?
What do you think about this solution?
We value your feedback to improve our textbook solutions.