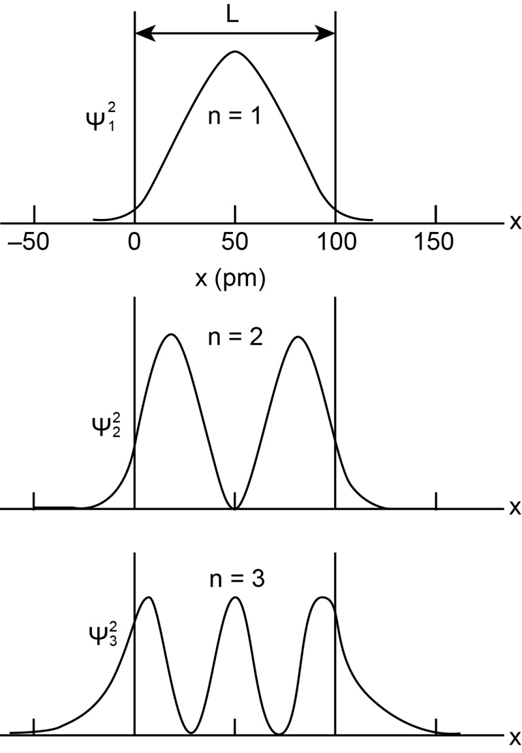
Chapter 39: Q24P (page 1216)
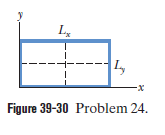
Figure 39-30 shows a two-dimensional, infinite-potential well lying in an xy plane that contains an electron. We probe for the electron along a line that bisects and find three points at which the detection probability is maximum. Those points are separated by 2.00 nm . Then we probe along a line that bisects and find five points at which the detection probability is maximum. Those points are separated by 3.00 nm . What is the energy of the electron?
Short Answer
The energy of the electron is 0.136 eV.