Chapter 39: Q23P (page 1216)
An electron is contained in the rectangular box of Fig. 39-14, with widths , , and . What is the electron’s ground-state energy?
Short Answer
The electron’s ground state energy is 3.2 eV.
Learning Materials
Features
Discover
Chapter 39: Q23P (page 1216)
An electron is contained in the rectangular box of Fig. 39-14, with widths , , and . What is the electron’s ground-state energy?
The electron’s ground state energy is 3.2 eV.
All the tools & learning materials you need for study success - in one app.
Get started for free
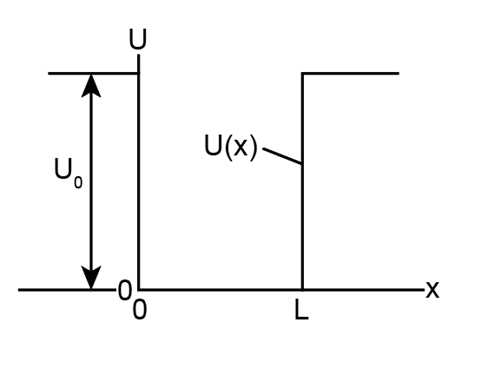
You want to modify the finite potential well of Fig. 39-7 to allow its trapped electron to exist in more than four quantum states. Could you do so by making the well (a) wider or narrower, (b) deeper or shallower?
Calculate the energy change required for an electron to move between states: a quantum jump up or down an energy-level diagram.
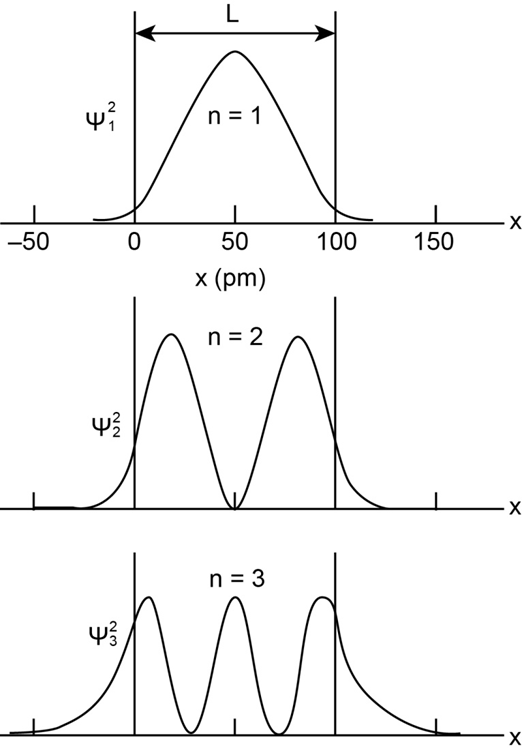
An electron is trapped in a one-dimensional infinite potential well in a state with quantum numbern = 17 . How many points of (a) zero probability and (b) maximum probability does its matter wave have?
From a visual inspection of Fig. 39-8, rank the quantum numbers of the three quantum states according to the de Broglie wavelength of the electron, greatest first.
Let be the energy difference between two adjacent energy levels for an electron trapped in a one-dimensional infinite potential well. Let E be the energy of either of the two levels. (a) Show that the ratio approaches the value at large values of the quantum number n. As , does (b) (c) E or, (d) approaches zero? (e) what do these results mean in terms of the correspondence principle?
What do you think about this solution?
We value your feedback to improve our textbook solutions.