Chapter 39: Q11Q (page 1214)
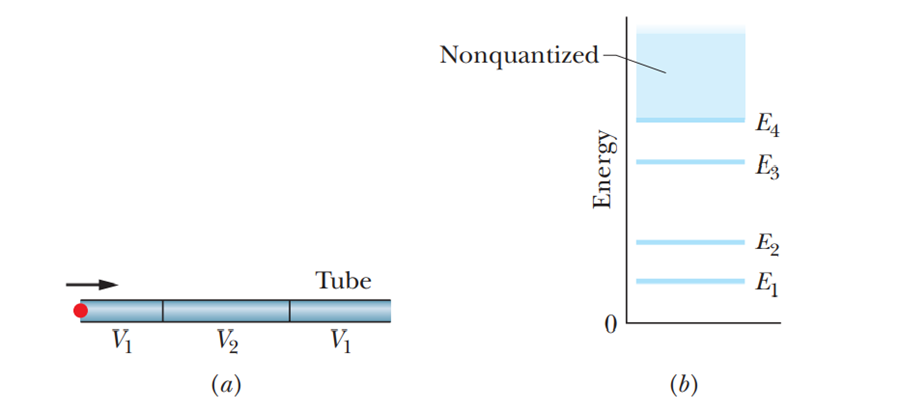
From a visual inspection of Fig. 39-8, rank the quantum numbers of the three quantum states according to the de Broglie wavelength of the electron, greatest first.

Short Answer
According to de Broglie wavelength, the electron rank of the quantum states with gratest first is n = 1, n = 2, n = 3.