Chapter 39: Q8Q (page 1214)
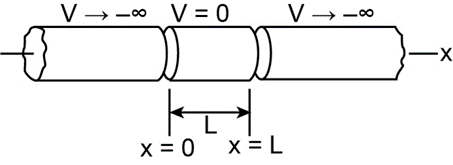
An electron is trapped in a finite potential well that is deep enough to allow the electron to exist in a state with n = 4. How many points of (a) zero probability and (b) maximum probability does its matter wave have within the well?
Short Answer
- The number of points of zero probability does its matter wave have is 5.
- The number of points of maximum probability does its matter wave have is 4.