Chapter 39: Q22P (page 1216)
An electron is contained in the rectangular corral of Fig. 39-13, with widths and. What is the electron’s ground-state energy?
Short Answer
The ground state energy of the electron is 0.7375 eV.
Learning Materials
Features
Discover
Chapter 39: Q22P (page 1216)
An electron is contained in the rectangular corral of Fig. 39-13, with widths and. What is the electron’s ground-state energy?
The ground state energy of the electron is 0.7375 eV.
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider an atomic nucleus to be equivalent to a one dimensional infinite potential well with , a typical nuclear diameter. What would be the ground-state energy of an electron if it were trapped in such a potential well? (Note: Nuclei do not contain electrons.)
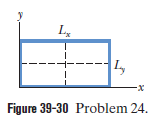
Figure 39-30 shows a two-dimensional, infinite-potential well lying in an xy plane that contains an electron. We probe for the electron along a line that bisects and find three points at which the detection probability is maximum. Those points are separated by 2.00 nm . Then we probe along a line that bisects and find five points at which the detection probability is maximum. Those points are separated by 3.00 nm . What is the energy of the electron?
Consider a conduction electron in a cubical crystal of a conducting material. Such an electron is free to move throughout the volume of the crystal but cannot escape to the outside. It is trapped in a three-dimensional infinite well. The electron can move in three dimensions so that its total energy is given by
in whichare positive integer values. Calculate the energies of the lowest five distinct states for a conduction electron moving in a cubical crystal of edge length .
particle is confined to the one-dimensional infinite potential well of Fig. 39-2. If the particle is in its ground state, what is its probability of detection between (a) , (b) , and
(c) ?
The two-dimensional, infinite corral of Fig. 39-31 is square, with edge length L = 150 pm. A square probe is centered at xy coordinates and has an x width of 5.00 pm and a y width of 5.00 pm . What is the probability of detection if the electron is in the energy state?

What do you think about this solution?
We value your feedback to improve our textbook solutions.