Chapter 5: Q15P (page 114)
Correlation coefficient and Excel graphing. Synthetic
data are given below for a calibration curve in which random Gaussian
noise with a standard deviation ofwas superimposed on
y values for the equation
. This exercise shows that
a high value of
does not guarantee that data quality is excellent.
(a)Enter concentration in column A and signal in column B of a
spreadsheet. Prepare an XY Scatter chart of signal versus concentration
without a line as described in Section
. Use LINEST
(Section 4-7) to find the least-squares parameters including
.
(b)Now insert the Trendline by following instructions on page 88.
In the Options window used to select the Trendline, select Display
Equation and Display R-Squared. Verify that Trendline and LINEST
give identical results.
(c)Add confidence interval y error bars following the instructions
at the end of Section 4-9. The
confidence interval is 6tsy,
where
comes from LINEST and Student’s tcomes from Table 4-4
forconfidence and
degrees of freedom. Also, compute
t with the statement .
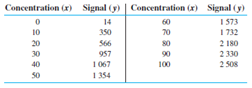
(a) The value of is and
(b) It is verified that Trendline displays the same data as LINEST.
(c) The confidence interval for y is calculated for the following bars has to be added and t with the statement " =" was determined.
Step by step solution
01
Definition linest and linearity
Linest : The linest is a Microsoft Excel function that measures the statistics for a straight line and an array defining that line using the least squares approach. It is a built-in function that may be categorised. Linearity: It's a measurement of how closely a calibration curve follows a straight line, indicating that the response is proportionate to the analyte quantity.
02
Find the least-squares parameters
(a)
Plot the graph with x as concentration values and y with signal values.
The value of
03
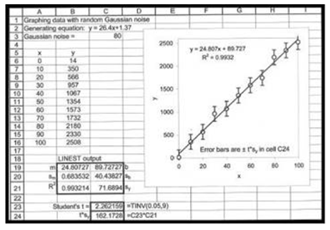
Check the trendline and linest
(b)
Again consider the graph with x as concentration values and y with signal values.
The Trendline displays the same data as LINEST, which are displayed within the graph.
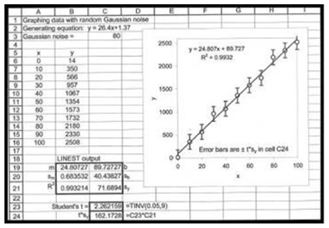
04
Determine t
(c)
The graph with x as concentration values and y with signal values.
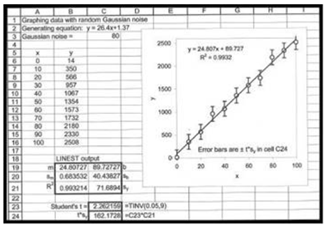
Therefore,confidence interval for is calculated in cell
Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their
learning with 91Ӱ��!





