Chapter 18: Q4TY (page 442)
If the observed absorbance is 0.200 and the blank absorbance is 0.049,
what is the concentration of Fe (mg/mL) in the serum?
Short Answer
The concentration of Fe (mg/mL) in the serum is
Learning Materials
Features
Discover
Chapter 18: Q4TY (page 442)
If the observed absorbance is 0.200 and the blank absorbance is 0.049,
what is the concentration of Fe (mg/mL) in the serum?
The concentration of Fe (mg/mL) in the serum is
All the tools & learning materials you need for study success - in one app.
Get started for free
18-8: What is an absorption spectrum?
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite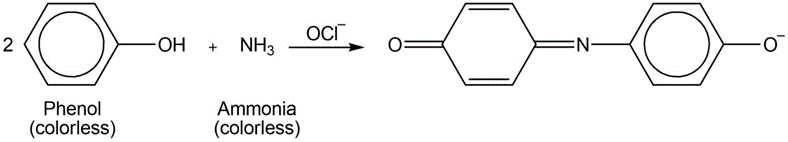
A 4.37 - mgsample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0mL. Thenof the solution were placed in a 50 - mLvolumetric flask and treated with 5mLof phenol solution plus 2mLof sodium hypochlorite solution. The sample was diluted to 50.0mLand the absorbance at 625nmwas measured in a 1.00 - cmcuvet after 30min. For reference, a standard solution was prepared from 0.0100g of dissolved in 1.00Lof water. Then 10.0mLof this standard were placed in a50 - mL volumetric flask and analyzed in the same manner as the unknown. A reagent blank was prepared by using distilled water in place of unknown.
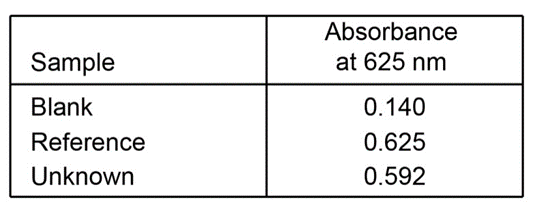
(a)Calculate the molar absorptivity of the blue product.
(b)Calculate the weight percent of nitrogen in the protein.
If a sample for spectrophotometric analysis is placed in a 10 - cmcell, the absorbance will be 10times greater than the absorbance in a 1 - cmcell. Will the absorbance of the reagent-blank solution also be increased by a factor of 10?
Nitrite ion, is a preservative for bacon and other foods, but it is potentially carcinogenic. A spectrophotometric determination ofmakes use of the following reactions
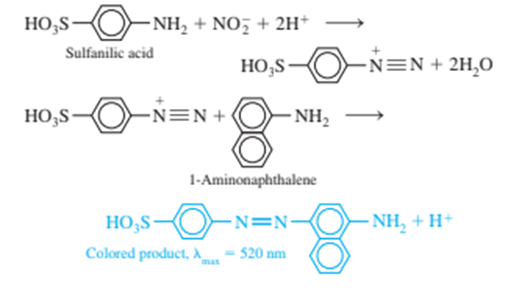
Here is an abbreviated procedure for the
determination:
1. To 50.0ml of unknown solution containing nitrite is added 1.00mL of sulfanilic acid solution.
2. After 10min , 2.00mL of 1 –a minonaphthalene solution and 1.00 mL of buffer are added.
3. After 15 min, the absorbance is read at 520 nm in a 5.00-cm cell.
The following solutions were analyzed:
A. 50.0 mL of food extract known to contain no nitrite (that is, a negligible amount); final absorbance =0.153.
B. 50.0mL of food extract suspected of containing nitrite; final absorbance \(=0.622\).
C. Same as B, but with added to the 50.0-mL sample; final absorbance =0.967.
(a) Calculate the molar absorptivity, of the colored product. Remember that a \(5.00\)-cm cell was used.
(b) How many micrograms ofwere present in 50.0mL of food extract?
Dilution by mass is more accurate than dilution by volume. A stocksolution contains 1.044 g Fe/kg solution in 0.48 M HCl. How many solution are in a solution made by mixing 2.145 g stock solution with 243.27 g 0.1 M HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.