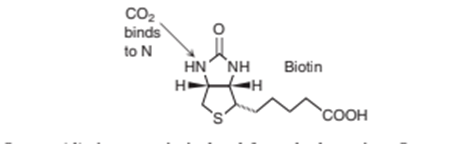
Chapter 18: QC E (page 455)
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite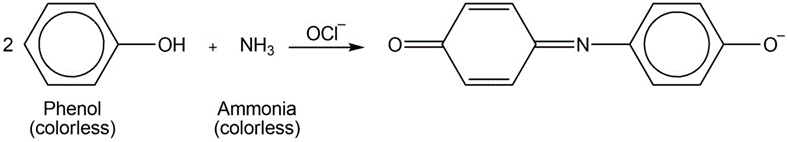
A 4.37 - mgsample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0mL. Thenof the solution were placed in a 50 - mLvolumetric flask and treated with 5mLof phenol solution plus 2mLof sodium hypochlorite solution. The sample was diluted to 50.0mLand the absorbance at 625nmwas measured in a 1.00 - cmcuvet after 30min. For reference, a standard solution was prepared from 0.0100g of dissolved in 1.00Lof water. Then 10.0mLof this standard were placed in a50 - mL volumetric flask and analyzed in the same manner as the unknown. A reagent blank was prepared by using distilled water in place of unknown.
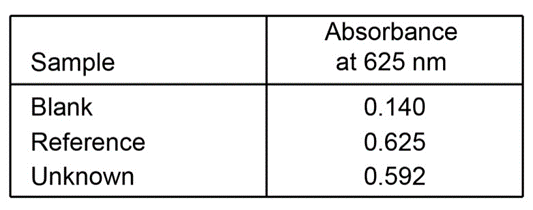
(a)Calculate the molar absorptivity of the blue product.
(b)Calculate the weight percent of nitrogen in the protein.
Short Answer
- Thevalue of the molar absorptivity =
- The weight of the nitrogen protein 16.1%