Chapter 18: Q7TY (page 444)
In a different titration, the absorbance after adding 75 mL of ferric nitrilo-triacetate to 1.500 mL of Apo transferrin was 0.222. Find the corrected absorbance.
Short Answer
The corrected absorbance is
Learning Materials
Features
Discover
Chapter 18: Q7TY (page 444)
In a different titration, the absorbance after adding 75 mL of ferric nitrilo-triacetate to 1.500 mL of Apo transferrin was 0.222. Find the corrected absorbance.
The corrected absorbance is
All the tools & learning materials you need for study success - in one app.
Get started for free
Preparing standards for a calibration curve.
(a) How much ferrous ammonium sulfateshould be dissolved in a 500mL volumetric flask withto obtain a stock solution with?
(b) When making stock solution (a), you weighed out 3.627 g of reagent. What is the Fe concentration in?
(c) How would you prepare 250mL of standard containing containing infrom stock solution (b) using only 5- and 10-mL Class A pipets, only 250mL volumetric flasks, and only two consecutive dilutions of the stock solution? For example, to prepare a solution with , you could first dilute 15mL(=10+5mL) of stock solution up to 250 mL Then dilute 15mL of the new solution up to 250 mL again to get
When I was a boy, Uncle Wilbur let me watch as he analyzed the iron content of runoff from his banana ranch. A 25.0 - mLsample was acidified with nitric acid and treated with excess KSCN to form a red complex. (KSCN itself iscolorless.) The solution was diluted to 100.0mLand put in a variable-pathlength cell. For comparison, a 10.0 - mLreference sample ofwas treated with andand diluted to. The reference was placed in a cell with a 1.00 - mL light path. The runoff sample exhibited the same absorbance as the reference when the path length of the runoff cell was 2.48cm. What was the concentration of iron in Uncle Wilbur’s runoff?
Vapor at a pressure of bar from the solid compound pyrazine had a transmittance of 24.4% at a wavelength of 266nm in
a 3.00 - cm cell at 298K
 Pyrazine
Pyrazine
(a) Convert transmittance to absorbance.
(b) Convert pressure to concentration (mol/L) with the ideal gas law (Problem 1-18).
(c) Find the molar absorptivity of gaseous pyrazine at 266nm.
18-29. Biotin-streptavidin fluorescence titration. Biotin is a cofactor in enzymatic carboxylation reactions. Biotin activatesfor biosynthetic reactions.
Streptavidin is a protein isolated from the bacterium Streptomyces avidinii that binds biotin with a formation constant of . The biotin-streptavidin complex is widely used in biotechnology because the noncovalent complex is stable in the presence of detergents, protein denaturants, and organic solvents, and at extremes of pH and temperature.
The stoichiometry of the biotin-streptavidin complex was measured by a fluorescence titration. Fluorescein (page 453 ) covalently attached to biotin via the biotin carboxyl group fluoresces at 520 nm when irradiated at 493 nm. When biotin-fluorescein (BF) binds to streptavidin (SA), fluorescence decreases. The table gives emission intensity for addition of BF to SA and also for addition of SA to BF. Data are already corrected for dilution.
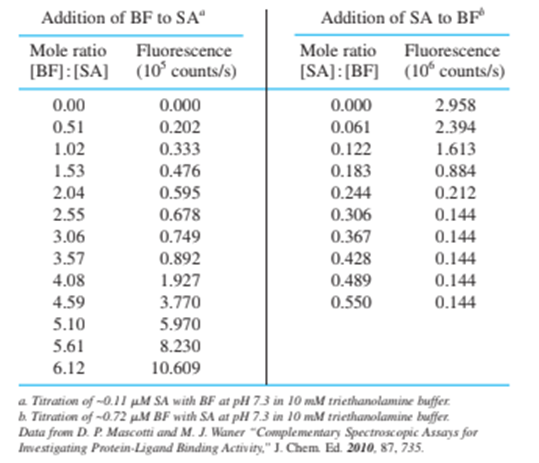
(a) Make a graph of fluorescence versus mole ratio for each titration and state the stoichiometry of binding of biotin to streptavidin.
(b) Explain the shape of each titration curve.
What is the difference between luminescence and Chemi-luminescence?
What do you think about this solution?
We value your feedback to improve our textbook solutions.