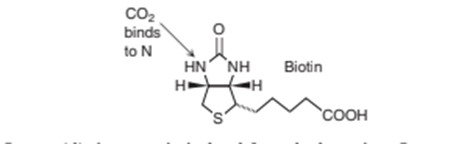
Chapter 18: Q14 P (page 456)
When I was a boy, Uncle Wilbur let me watch as he analyzed the iron content of runoff from his banana ranch. A 25.0 - mLsample was acidified with nitric acid and treated with excess KSCN to form a red complex. (KSCN itself iscolorless.) The solution was diluted to 100.0mLand put in a variable-pathlength cell. For comparison, a 10.0 - mLreference sample ofwas treated with andand diluted to. The reference was placed in a cell with a 1.00 - mL light path. The runoff sample exhibited the same absorbance as the reference when the path length of the runoff cell was 2.48cm. What was the concentration of iron in Uncle Wilbur’s runoff?
Short Answer
The concentration of iron in Uncle Wilbur’s runoff is role="math" localid="1663651601222" .