Chapter 18: Q1TY (page 460)
What is the wavelength, wavenumber, and name of radiation with an energy of 100 kJ/mol?
Short Answer
The wavelength is
The wavenumber is
The name of radiation is Infrared radiation.
Learning Materials
Features
Discover
Chapter 18: Q1TY (page 460)
What is the wavelength, wavenumber, and name of radiation with an energy of 100 kJ/mol?
The wavelength is
The wavenumber is
The name of radiation is Infrared radiation.
All the tools & learning materials you need for study success - in one app.
Get started for free
Vapor at a pressure of bar from the solid compound pyrazine had a transmittance of 24.4% at a wavelength of 266nm in
a 3.00 - cm cell at 298K
 Pyrazine
Pyrazine
(a) Convert transmittance to absorbance.
(b) Convert pressure to concentration (mol/L) with the ideal gas law (Problem 1-18).
(c) Find the molar absorptivity of gaseous pyrazine at 266nm.
Gold nanoparticles (Figure 17-31) can be titrated with the oxidizing agent TCNQ in the presence of excess ofto oxidize in deaerated toluene. Gold atoms in the interior of the particle are Au(0) . Gold atoms bound to - (dodecanethiol) ligands on the surface of the particle are Au(I) and are not titrated.
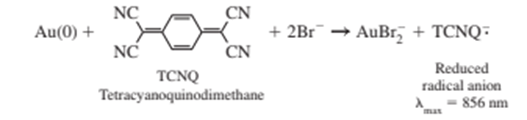
The table gives the absorbance at $856 0.700 mL of in toluene is titrated with gold nanoparticles (1.43 g/Lin toluene) from a microsyringe with a Teflon-coated needle. Absorbance in the table has already been corrected for dilution.
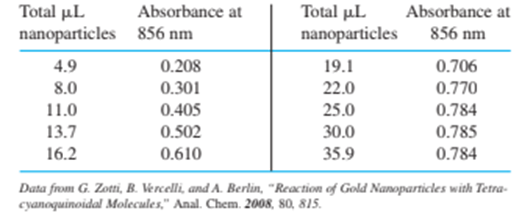
(a) Make a graph of absorbance versus volume of titrant and estimate the equivalence point. Calculate the Au(0) in 1.00 g of nanoparticles.
(b) From other analyses of similarly prepared nanoparticles, it is estimated that 25 % of the mass of the particle is dodecanethiol ligand. Calculate mmol of localid="1667559229564" in 1.00 g of nanoparticles.
(c) The Au(I) content of 1.00 g of nanoparticles should be 1.00 mass of Au(0) - mass of . Calculate the micromoles of Au(I) in 1.00 g of nanoparticles and the mole ratio Au(I) :. In principle, this ratio should be 1: 1. The difference is most likely because was not measured for this specific nanoparticle preparation.
In formaldehyde, the transition occurs at 397 nm,and the transition comes at 355 nm. What is the difference in energy (kJ/mol) between the andstates? This difference is due to the different electron spins in the two states.
Describe a different set of dilutions to produce 3 mg Fe/mL from 1 000 mg
Fe/mL
Dilution by mass is more accurate than dilution by volume. A stocksolution contains 1.044 g Fe/kg solution in 0.48 M HCl. How many solution are in a solution made by mixing 2.145 g stock solution with 243.27 g 0.1 M HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.