Chapter 18: Q6TY (page 443)
Describe a different set of dilutions to produce 3 mg Fe/mL from 1 000 mg
Fe/mL
Short Answer
Dilute 10 mL of stock to 100 mL to get. Dilute of the new solution up to 100 mL to get
Learning Materials
Features
Discover
Chapter 18: Q6TY (page 443)
Describe a different set of dilutions to produce 3 mg Fe/mL from 1 000 mg
Fe/mL
Dilute 10 mL of stock to 100 mL to get. Dilute of the new solution up to 100 mL to get
All the tools & learning materials you need for study success - in one app.
Get started for free
In formaldehyde, the transition occurs at 397 nm,and the transition comes at 355 nm. What is the difference in energy (kJ/mol) between the andstates? This difference is due to the different electron spins in the two states.
(a) How much energy (in kilojoules) is carried by one mole of photons of red light with= 650 nm?
(b) How many kilojoules are carried by one mole of photons of violet light with= 400 nm?
A compound with molecular mass 292.16g/molwas dissolved in a 5 - mLvolumetric flask. A1.00 - mLaliquot was withdrawn, placed in a 10 - mLvolumetric flask, and diluted to the mark. The absorbance atwas in 0.427 i acuvet. The molar absorptivity at 340nm is
(a) Calculate the concentration of compound in the cuvet.
(b) What was the concentration of compound in the 5 - mL flask?
(c) How many milligrams of compound were used to make the 5 - mLsolution?
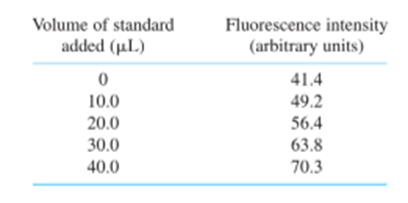
Standard addition. Selenium from 0.108 g of Brazil nuts was converted into the fluorescent product in Reaction 18-15, and extracted into 10.0 mL of cyclohexane. Then 2.00 mL of the cyclohexane solution were placed in a cuvet for fluorescence measurement. Standard additions of fluorescent product containingSe/mL are given in the table. Construct a standard addition graph to find the concentration of Se in the 2.00-mL unknown solution. Find the wt%of Se in the nuts and its uncertainty and 95% confidence interval
What is the wavelength, wavenumber, and name of radiation with an energy of 100 kJ/mol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.