Chapter 18: QE E (page 456)
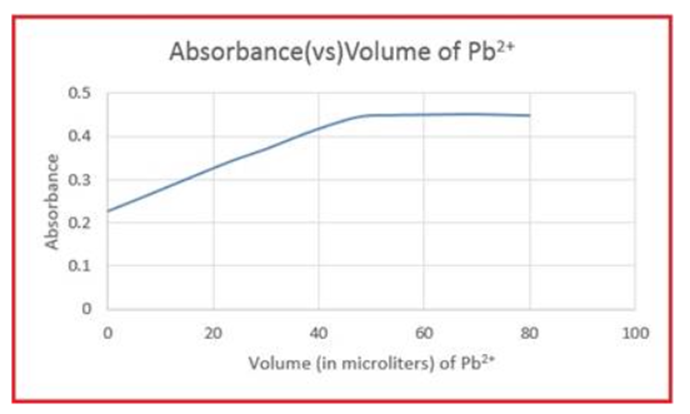
Semi-xylenol orange is a yellow compound at pH 5.9but turns red when it reacts with . A 2.025 - mLsample of semixylenol orange at pH 5.9was titrated withwith the following results:
Total role="math" localid="1663647483742" added | Absorbance at 490nmwavelength | Total added | Absorbance at490nmwavelength |
0.0 6.0 12.0 18.0 24.0 30.0 36.0 | 0.227 0.256 0.286 0.316 0.345 0.370 0.399 | 42.0 48.0 54.0 60.0 70.0 80.0 | 0.425 0.445 0.448 0.449 0.450 0.447 |
Make a graph of absorbance versus microliters of added. Be sure to correct the absorbances for dilution. Corrected absorbance is what would be observed if the volume were not changed from its initial value of 2.025Ml. Assuming that the reaction of semi orange with has a 1 : 1stoichiometry, find the molarity of semi-xylenol orange in the original solution.
Short Answer
The molarity of semi-xylenol is