18-29. Biotin-streptavidin fluorescence titration. Biotin is a cofactor in enzymatic carboxylation reactions. Biotin activatesfor biosynthetic reactions.
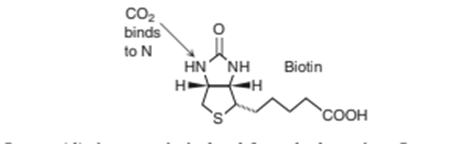
Streptavidin is a protein isolated from the bacterium Streptomyces avidinii that binds biotin with a formation constant of . The biotin-streptavidin complex is widely used in biotechnology because the noncovalent complex is stable in the presence of detergents, protein denaturants, and organic solvents, and at extremes of pH and temperature.
The stoichiometry of the biotin-streptavidin complex was measured by a fluorescence titration. Fluorescein (page 453 ) covalently attached to biotin via the biotin carboxyl group fluoresces at 520 nm when irradiated at 493 nm. When biotin-fluorescein (BF) binds to streptavidin (SA), fluorescence decreases. The table gives emission intensity for addition of BF to SA and also for addition of SA to BF. Data are already corrected for dilution.
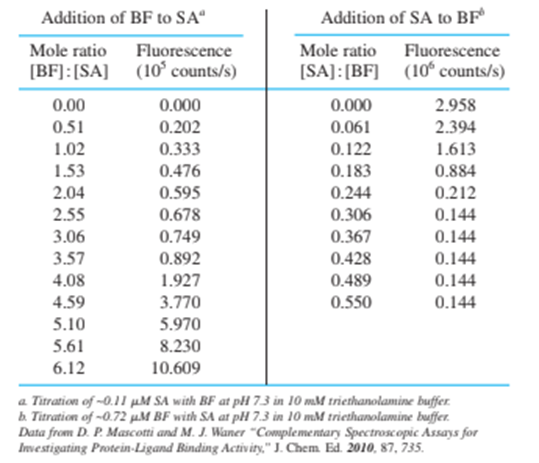
(a) Make a graph of fluorescence versus mole ratio for each titration and state the stoichiometry of binding of biotin to streptavidin.
(b) Explain the shape of each titration curve.




