Chapter 6: Q41P (page 143)
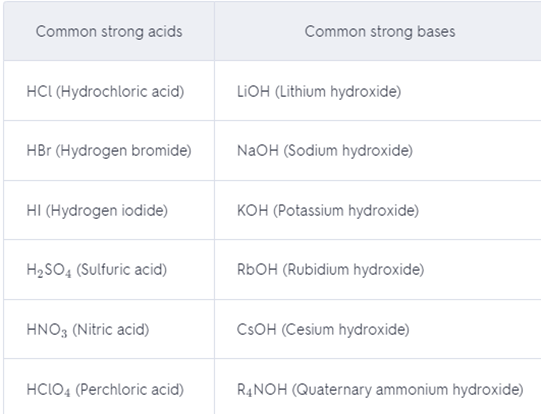
Make a list of the common strong acids and strong bases. Memorize this list.
Short Answer
The list:
Learning Materials
Features
Discover
Chapter 6: Q41P (page 143)
Make a list of the common strong acids and strong bases. Memorize this list.
The list:

All the tools & learning materials you need for study success - in one app.
Get started for free
Henry's law states that the concentration of a gas dissolved in a liquid is proportional to the pressure of the gas. This law is a consequence of the equilibrium
where is called the Henry's law constant. For the gasoline additive MTBE, . Suppose we have a closed container with aqueous solution and air in equilibrium. If the concentration of MTBE in the water is , what is the pressure of MTBE in the air?
Methyl- -butyl ether (MTBE, FM 88.15)
A solution contains and . Can 99.90% of either ion be precipitated by chromate without precipitating the other metal ion?
Reaction 6-8 is allowed to come to equilibrium in a solution initially containing, and 1.00MH+. To find the concentrations at equilibrium, we construct the table at the bottom of the page showing initial and final concentrations. We use the stoichiometry coefficients of the reaction to say that if of are created, then we must also make x mol of and 8x mol of H+. To produce x mol of , we must have consumed x mol of and 2x mol of Cr3+.
(a) Write the equilibrium constant expression that you would use to solve for x to find the concentrations at equilibrium. Do not try to solve the equation.
(b) Because , we suppose that the reaction will go nearly "to completion." That is, we expect both the concentration of and to be close to 0.00500M an equilibrium. (Why?) That is, . With this value of and . However, we cannot say , because there must be some small concentration of at equilibrium. Write for the concentration of and solve for . The limiting reagent in this example is . The reaction uses up before consuming .
What concentration of (ferrocyanide) is in equilibrium with androle="math" localid="1663331502441" ? Express your answer with a prefix from Table 1-3.
Question: (a) From Kw in Table 6-1, calculate the pH of pure water at 00,200, and 400C.
(b) For the reaction , at 250C. In this equation, D stands for deuterium, which is the isotope 2H. What is the pD (=-log[D+]) for neutral D2O?
What do you think about this solution?
We value your feedback to improve our textbook solutions.