Chapter 6: Q20P (page 142)
A solution contains and . Can 99.90% of either ion be precipitated by chromate without precipitating the other metal ion?
Short Answer
Neither norcan be precipitated without precipitating other metal ion.
Learning Materials
Features
Discover
Chapter 6: Q20P (page 142)
A solution contains and . Can 99.90% of either ion be precipitated by chromate without precipitating the other metal ion?
Neither norcan be precipitated without precipitating other metal ion.
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify the Bronsted-Lowry acids among the reactants in the following reactions:
(a)
(b)
for dimethylamine is . Find for dimethyl ammonium ion.
Write the autoprotolysis reaction of.
From the following equilibrium constants, calculate the equilibrium constant for the reaction
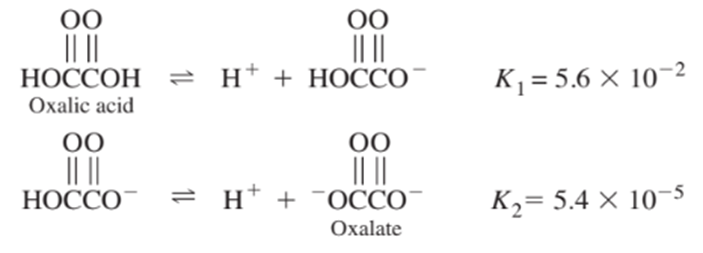
Calculate the pHof pure water at (a) and (b).
What do you think about this solution?
We value your feedback to improve our textbook solutions.