Chapter 6: Q52P (page 143)
From the following equilibrium constants, calculate the equilibrium constant for the reaction
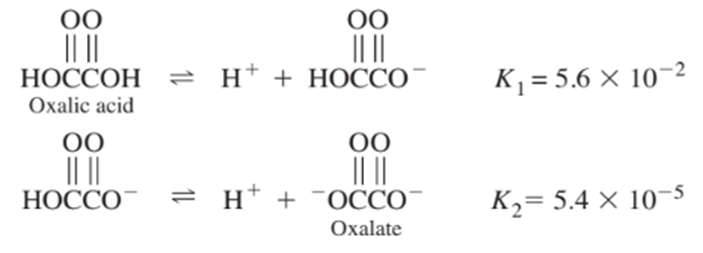
Short Answer
Equilibrium constant (K) for given reaction was calculated as
Learning Materials
Features
Discover
Chapter 6: Q52P (page 143)
From the following equilibrium constants, calculate the equilibrium constant for the reaction

Equilibrium constant (K) for given reaction was calculated as
All the tools & learning materials you need for study success - in one app.
Get started for free
For the reaction , the concentrations at equilibrium are found to be
A:
B:
C:
D:
E:
Find the numerical value of the equilibrium constant that would appear in a conventional table of equilibrium constants.
Use electron dot structures to show why tetra methyl ammonium hydroxide, , is an ionic compound. That is, show why hydroxide is not covalently bound to the rest of the molecule.
Calculate andpHfor the following solutions:
(a)
(b)
(c)
(d)
(e)
Tetramethylammonium hydroxide
Distinguish Lewis acids and bases from Brønsted-Lowry acids and bases. Give an example of each.
(a) A favorable entropy change occurs when is positive. Does the order of the system increase or decrease when is positive?
(b) A favorable enthalpy change occurs when is negative. Does the system absorb heat or give off heat when is negative?
(c) Write the relation between ,and . Use the results of parts (a) and (b) to state whether must be positive or negative for a spontaneous change.
What do you think about this solution?
We value your feedback to improve our textbook solutions.