Chapter 6: Q34P (page 143)
Write the autoprotolysis reaction of.
Short Answer
The autoprotolysis reaction for is:
role="math" localid="1663387364297"
Learning Materials
Features
Discover
Chapter 6: Q34P (page 143)
Write the autoprotolysis reaction of.
The autoprotolysis reaction for is:
role="math" localid="1663387364297"
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Histidine is a triprotic amino acid:
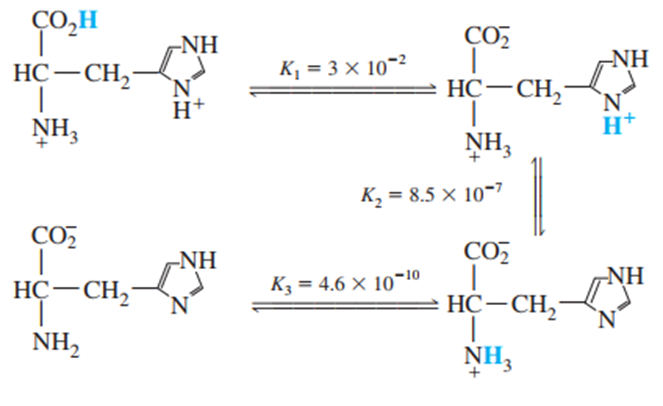
Find the value of the equilibrium constant for the reaction
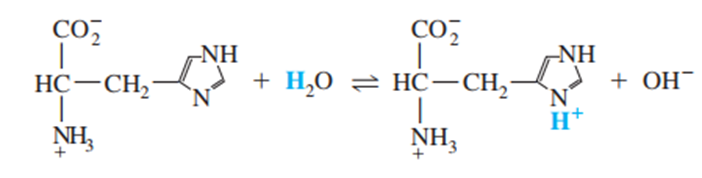
Find in equilibrium with CuBr(s) and 0.10MB.
: Write thereaction for pyridine and for sodium 2-mercaptoethanol.

Use electron dot structures to show why tetra methyl ammonium hydroxide, , is an ionic compound. That is, show why hydroxide is not covalently bound to the rest of the molecule.
loses water when it is heated in an oven:
a) Write the equilibrium constant for this reaction. Calculate the vapour pressure of gaseous above at 298K.
(b) If and are not temperature dependent (a poor assumption), estimate the temperature at which above will be 1 bar.
What do you think about this solution?
We value your feedback to improve our textbook solutions.