Chapter 6: QJ-E (page 141)
Question: Histidine is a triprotic amino acid:
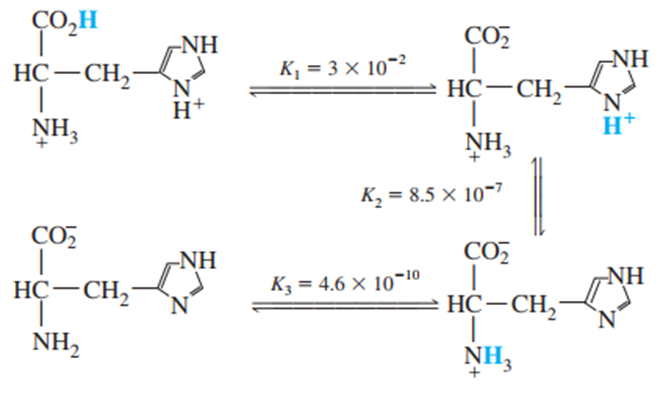
Find the value of the equilibrium constant for the reaction
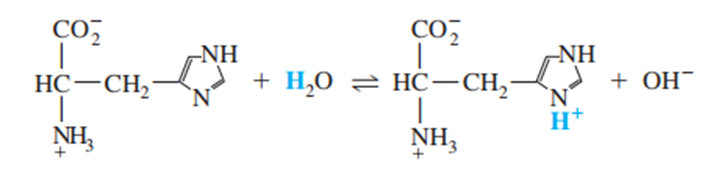
Short Answer
The equilibrium constant for the reaction is 1.17×10-8
Learning Materials
Features
Discover
Chapter 6: QJ-E (page 141)
Question: Histidine is a triprotic amino acid:

Find the value of the equilibrium constant for the reaction
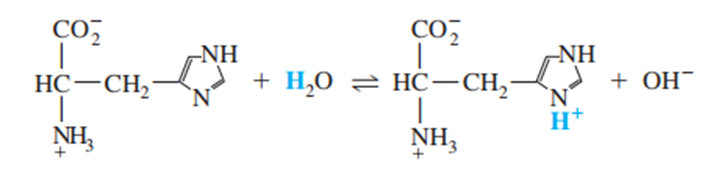
The equilibrium constant for the reaction is 1.17×10-8
All the tools & learning materials you need for study success - in one app.
Get started for free
From the following equilibrium constants, calculate the equilibrium constant for the reaction
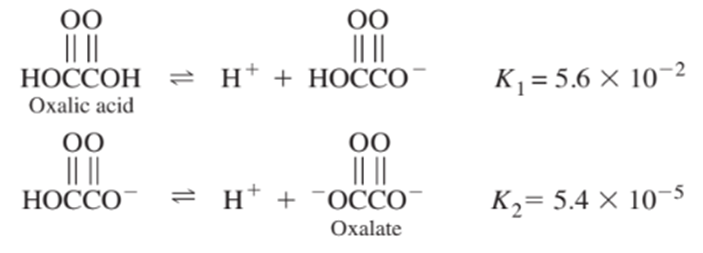
for chloroacetic acid is Find for chloroacetate ion.
for dimethylamine is . Find for dimethyl ammonium ion.
Find in the solution when excess solid lanthanum iodate, , is stirred with until the system reaches equilibrium. Assume that from is negligible compared with from .
(a) From the solubility product of zinc ferrocyanide, , calculate the concentration of in 0.1 0m M saturated with . Assume that is a negligible source of .
(b) What concentration of should be in a suspension of solid in water to give?
What do you think about this solution?
We value your feedback to improve our textbook solutions.