Chapter 6: Q30P (page 143)
Why is the pH of distilled water usually <7? How can you prevent this from happening?
Short Answer
The pH of distilled water usually <7 due to reaction of dissolved carbon dioxide with water.
Learning Materials
Features
Discover
Chapter 6: Q30P (page 143)
Why is the pH of distilled water usually <7? How can you prevent this from happening?
The pH of distilled water usually <7 due to reaction of dissolved carbon dioxide with water.
All the tools & learning materials you need for study success - in one app.
Get started for free
A solution contains and . Can 99.90% of either ion be precipitated by chromate without precipitating the other metal ion?
What concentration of (ferrocyanide) is in equilibrium with androle="math" localid="1663331502441" ? Express your answer with a prefix from Table 1-3.
From the following equilibrium constants, calculate the equilibrium constant for the reaction
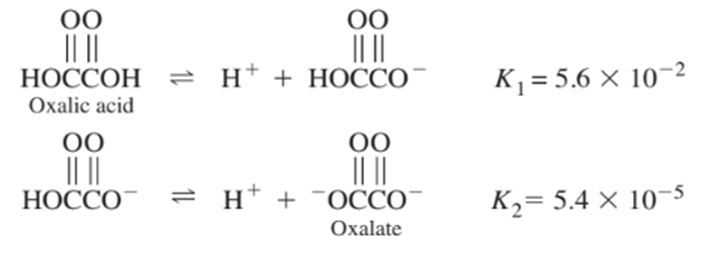
Question: (a) From Kw in Table 6-1, calculate the pH of pure water at 00,200, and 400C.
(b) For the reaction , at 250C. In this equation, D stands for deuterium, which is the isotope 2H. What is the pD (=-log[D+]) for neutral D2O?
Write the reaction of Given that the value for is calculate for.
What do you think about this solution?
We value your feedback to improve our textbook solutions.