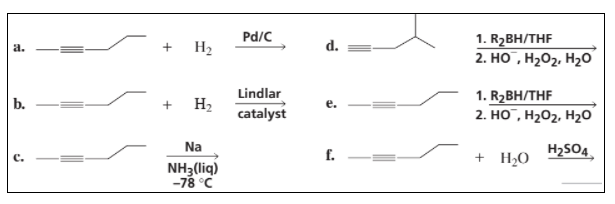
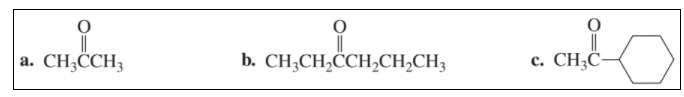
Chapter 7: Q8P (page 294)
What orbitals are used to form the carbon–carbonσbond between the highlighted carbons?
Short Answer
a) C-1 =sp2 b) C-2 = sp2 c) C-2 = sp d) C-2 =sp e) C-1 =sp
C-2 = sp2 C-3 = sp3 C-3 = sp2C-3 =sp3 C-2 =sp
f) C-2 =sp2 g) C-2 =sp2 h) C-2 =sp2i) C-1 =sp2
C-3 =sp2 C-3 =sp2 C-3 =sp3C-2 =sp