Chapter 20: Q67P (page 985)
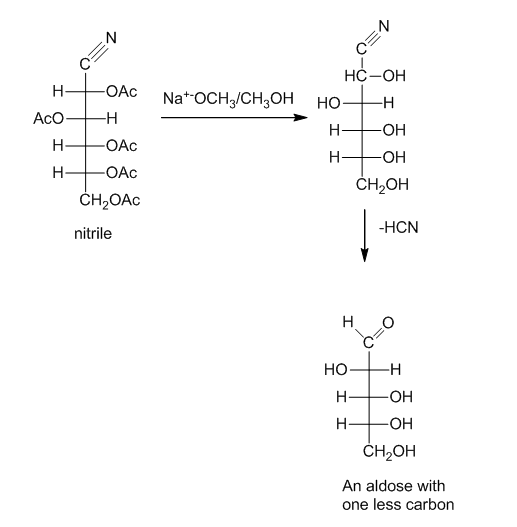
Draw the mechanism for the dehydration step in the Wohl degradation
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q67P (page 985)
Draw the mechanism for the dehydration step in the Wohl degradation

All the tools & learning materials you need for study success - in one app.
Get started for free
a. What sugar is the C-3 epimer of D-xylose?
b. What sugar is the C-5 epimer of D-allose?
c. What sugar is the C-4 epimer of L-gulose?
d. What sugar is the C-4 epimer of D-lyxose?
The 1H NMR spectrum of D- Glucose in D2O exhibits two high frequency doublets. What is responsible for these doublets?
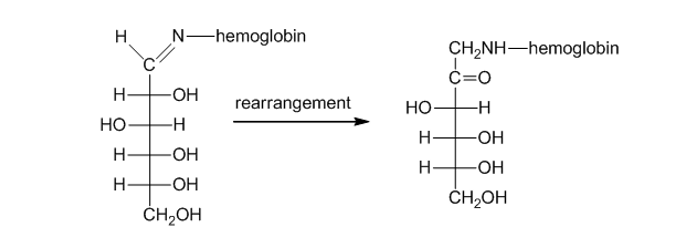
Propose a mechanism for the rearrangement that converts an -hydroxyimine to an α -aminoketone in the presence of a trace amount of acid (page 960).
What are the systematic names of the following compounds? Indicate the configuration (R or S) of each asymmetric center.a. D-glucose b. D-mannose c. D-galactosed. L-glucose
To synthesize D-galactose, a student went to the stockroom to get some D-lyxose to use as a starting material. She found that the labels had fallen off the bottles containing D-lyxose and D-xylose. How can she determine which bottle contains D-lyxose?
What do you think about this solution?
We value your feedback to improve our textbook solutions.