Chapter 20: Q-20-71P (page 985)
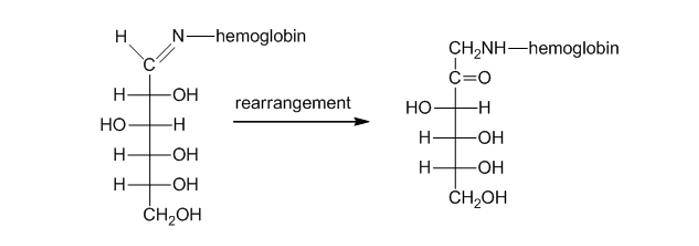
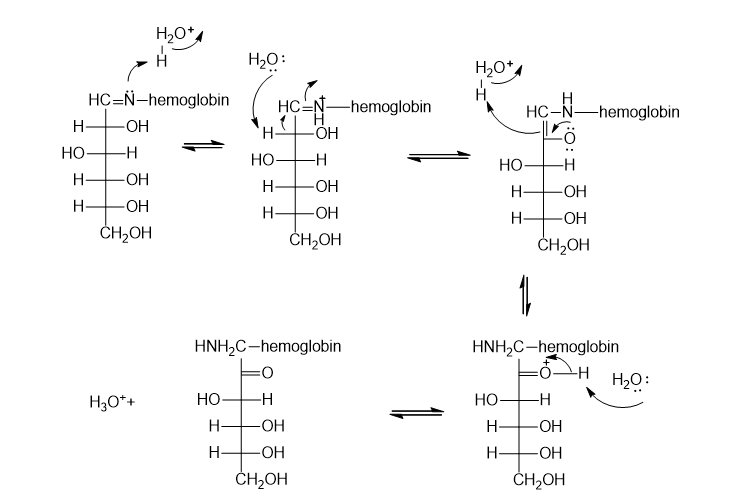
Propose a mechanism for the rearrangement that converts an -hydroxyimine to an α -aminoketone in the presence of a trace amount of acid (page 960).
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q-20-71P (page 985)
Propose a mechanism for the rearrangement that converts an -hydroxyimine to an α -aminoketone in the presence of a trace amount of acid (page 960).


All the tools & learning materials you need for study success - in one app.
Get started for free
All the glucose units in dextran have six-membered rings. When a sample of dextran is treated with methyl iodide and AgO and the product is hydrolyzed
under acidic conditions, the final products are 2,3,4,6-tetra-O-methyl-d-glucose, 2,4,6-tri-O-methyl-d-glucose, 2,3,4-tri-O-methyl-d-glucose, and 2,4-di-O-methyl-d-glucose. Draw a short segment of dextran.
Predict whether d-altrose exists preferentially as a pyranose or a furanose.
What sugar is the C-3 epimer of D-fructose?
Draw the mechanism for the formation of β-lactose from a α- D-galactose and β- D-glucose in dil HCl.
How many aldaric acids are obtained from the 16 aldohexoses?
What do you think about this solution?
We value your feedback to improve our textbook solutions.