Chapter 27: Q18P (page 1202)
What happens to polyester slacks if aqueous NaOH is spilled on them?
Short Answer
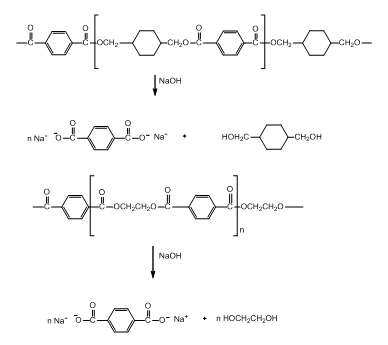
The polyester slacks hydrolyze to produce salt of dicarboxylic acid and diols.
The reaction is given below.
Learning Materials
Features
Discover
Chapter 27: Q18P (page 1202)
What happens to polyester slacks if aqueous NaOH is spilled on them?
The polyester slacks hydrolyze to produce salt of dicarboxylic acid and diols.
The reaction is given below.

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Draw three segments of the polymer that is formed from 1,4-polymerization of 1,3-butadiene in which all the double bonds are trans.
b. Draw three segments of the polymer that is formed from 1,2-polymerization of 1,3-butadiene.
Draw the product of each of the following reactions:

Propose a mechanism for the formation of Melmac.
Polyethylene is used for the production of beach chairs as well as beach balls. Which of these items is made from more highly branched polyethylene?
Show the mechanism for the formation of a segment of poly(vinyl chloride) that contains three units of vinyl chloride and is initiated by hydrogen peroxide.
What do you think about this solution?
We value your feedback to improve our textbook solutions.