Chapter 27: Q24P (page 1211)
Draw the product of each of the following reactions:

Short Answer
(a)
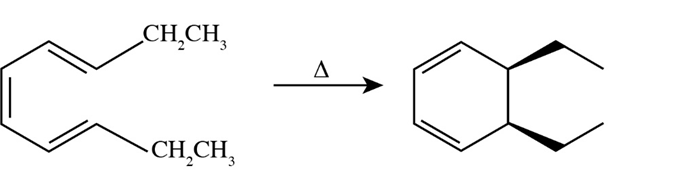
(b)
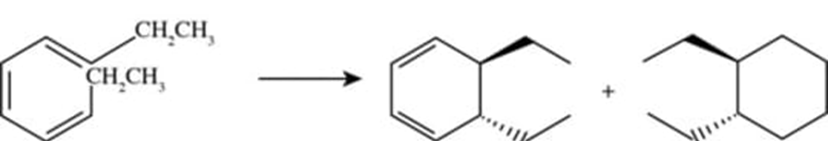
(c)
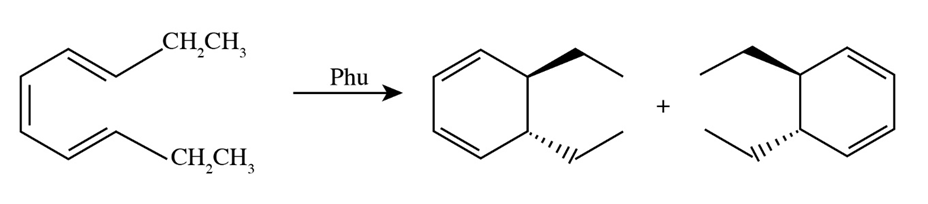
(d)
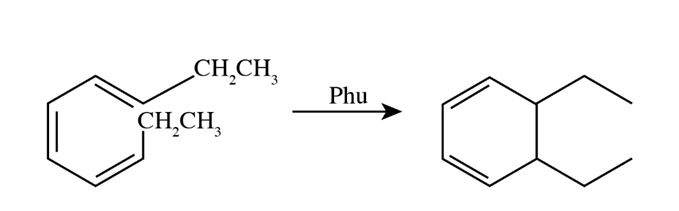
Learning Materials
Features
Discover
Chapter 27: Q24P (page 1211)
Draw the product of each of the following reactions:

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show the mechanism for the formation of a segment of poly(vinyl chloride) that contains three units of vinyl chloride and is initiated by hydrogen peroxide.
What monomer would you use to form each of the following polymers?
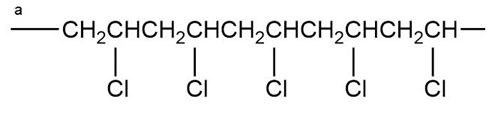
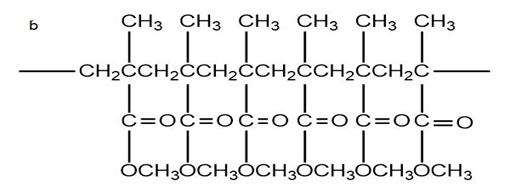
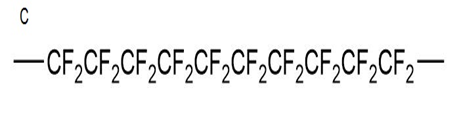
Explain why, when propylene oxide undergoes anionic polymerization, nucleophilic attack occurs at the less substituted carbon of the epoxide, but when it undergoes cationic polymerization, nucleophilic attack occurs at the more substituted carbon.
A particularly strong and rigid polyester used for electronic parts is marketed under the trade name Glyptal. It is a polymer of terephthalic acid and glycerol. Draw a segment of the polymer and explain why it is so strong.
Five different repeating units are found in the polymer obtained by cationic polymerisation of 4- methyl-1-pentene. Identify these repeating units.
What do you think about this solution?
We value your feedback to improve our textbook solutions.