Chapter 9: Q94P (page 391)
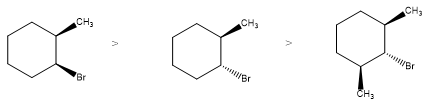
Rank the following from most reactive to least reactive in an E2reaction:
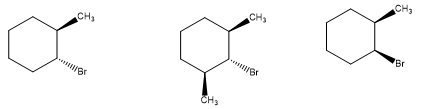
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q94P (page 391)
Rank the following from most reactive to least reactive in an E2reaction:


All the tools & learning materials you need for study success - in one app.
Get started for free
Which substitution reaction takes place more rapidly?
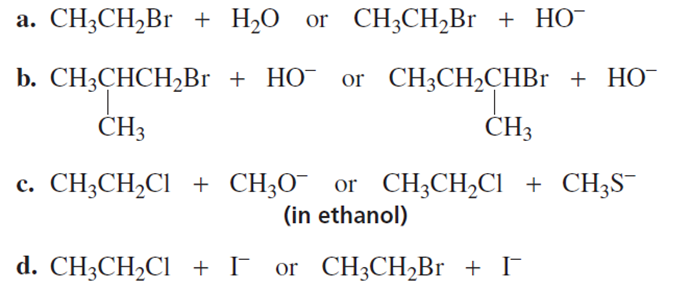
Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with
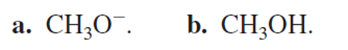
Would you expect acetate ion (CH3CO2-) to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxide?
The reaction of chloromethane with hydroxide ion at 30°C has a ΔG° value of -21.7 kcal/mol. What is the equilibrium constant for the reaction?
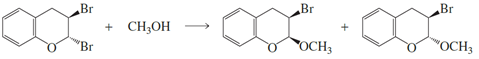
a. Propose a mechanism for the following reaction.
b. Explain why two products are formed.
c. Explain why methanol substitutes for only one of the bromines.
What do you think about this solution?
We value your feedback to improve our textbook solutions.