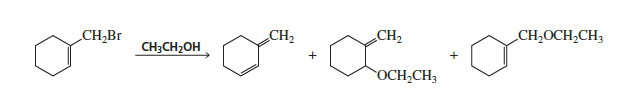
Chapter 9: Q58P (page 442)
Would you expect acetate ion (CH3CO2-) to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxide?
Short Answer
Yes,acetate ion (CH3CO2-) to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxide.