Chapter 9: Q56P (page 442)
Question: Which reaction in each of the following pairs takes place more rapidly? (EtOH is ethyl alcohol; Et2O is diethyl ether.)
Short Answer
Reaction in each of the following pairs takes place more rapidly are -
Learning Materials
Features
Discover
Chapter 9: Q56P (page 442)
Question: Which reaction in each of the following pairs takes place more rapidly? (EtOH is ethyl alcohol; Et2O is diethyl ether.)
Reaction in each of the following pairs takes place more rapidly are -
All the tools & learning materials you need for study success - in one app.
Get started for free
Which member in each pair in Problem 68 is a better leaving group?
Draw the products of the following intramolecular reactions:
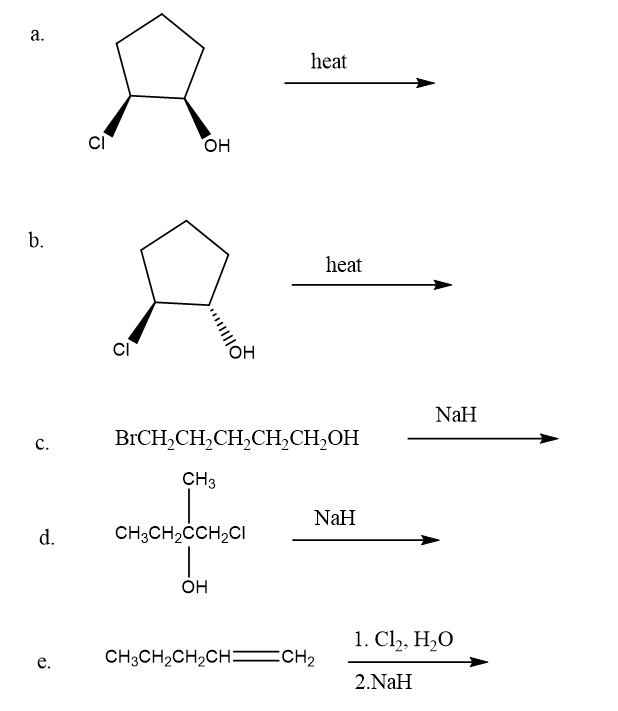
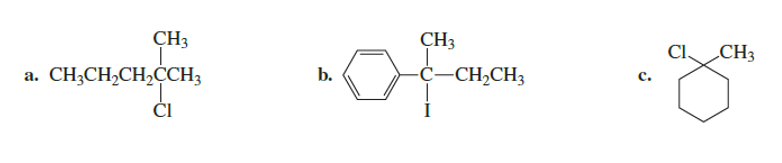
What is the major product formed when the following compounds undergo an E1 reaction?
What is the product of the reaction of bromoethane with each of the following nucleophiles?
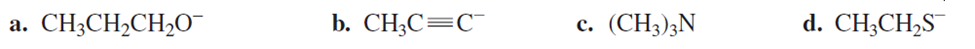
a. Which reacts faster in an E2 reaction: 3-bromocyclohexene or Bromo cyclohexane?
b. Which reacts faster in an E1 reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.