Chapter 9: Q92P (page 391)
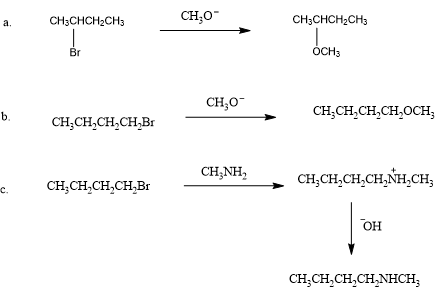
Starting with an alkyl halide, how could the following compounds be prepared?
a. 2-methoxybutane
b. 1-methoxybutane
c. butyl methylamine
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q92P (page 391)
Starting with an alkyl halide, how could the following compounds be prepared?
a. 2-methoxybutane
b. 1-methoxybutane
c. butyl methylamine

All the tools & learning materials you need for study success - in one app.
Get started for free
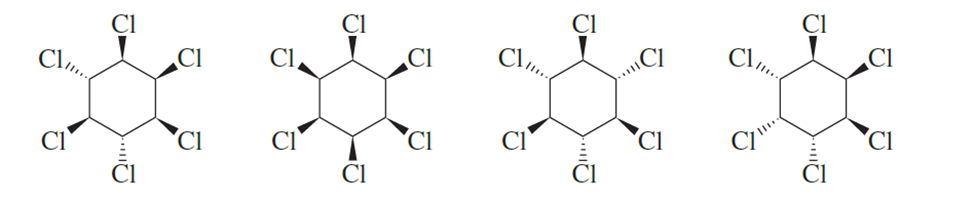
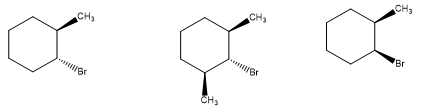
Which of the following hexachlorocyclohexanes is the least reactive in an E2 reaction?
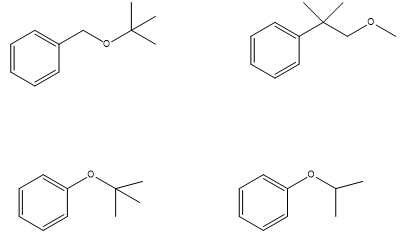
Which of following ethers cannot be made by a Williamson ether synthesis?
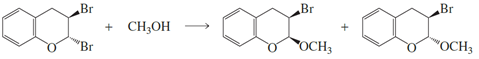
a. Propose a mechanism for the following reaction.
b. Explain why two products are formed.
c. Explain why methanol substitutes for only one of the bromines.
Why is a cumulated diene not formed in the reaction shown below?
Rank the following from most reactive to least reactive in an E2reaction:
What do you think about this solution?
We value your feedback to improve our textbook solutions.