Chapter 9: Q50P (page 436)
Why is a cumulated diene not formed in the reaction shown below?
Short Answer
Cumulative diene is not formed due to the instability in its transition state.
Learning Materials
Features
Discover
Chapter 9: Q50P (page 436)
Why is a cumulated diene not formed in the reaction shown below?
Cumulative diene is not formed due to the instability in its transition state.
All the tools & learning materials you need for study success - in one app.
Get started for free
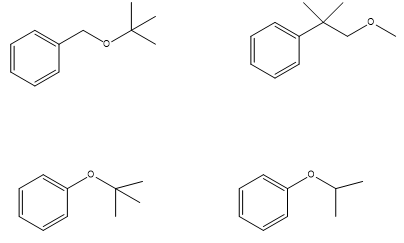
Which of following ethers cannot be made by a Williamson ether synthesis?
What product is obtained when the following compound undergoes two successive elimination reactions?

Does increasing the energy barrier for an SN2 reaction increase or decrease the magnitude of the rate constant for the reaction?
If 2-fluoropentane were to undergo an E1 reaction, would you expect the major product to be the more stable alkene or the less stable alkene? Explain your answer.
Explain why only a substitution product and no elimination product is obtained when the following Compound reacts with sodium methoxide:

What do you think about this solution?
We value your feedback to improve our textbook solutions.