Chapter 9: Q62P (page 444)
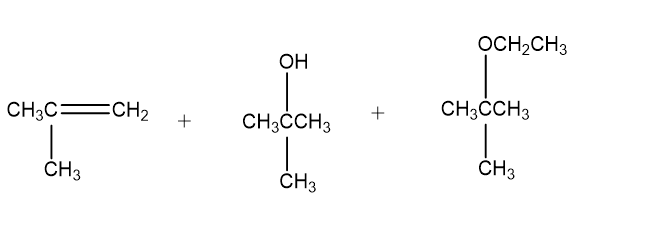
Identify the three products formed when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q62P (page 444)
Identify the three products formed when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water

All the tools & learning materials you need for study success - in one app.
Get started for free
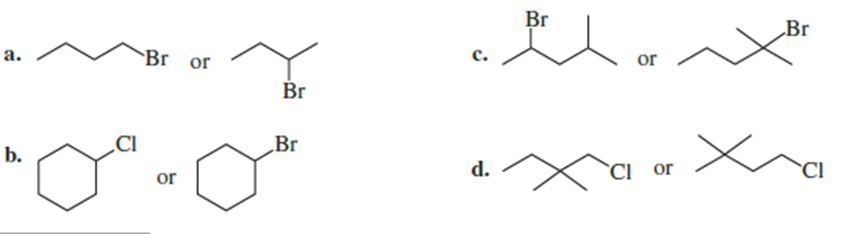
Which alkyl halide in each pair is more reactive in an E2 reaction with hydroxide ion?
Draw the substitution product formed by each of the following SN2 reactions:
a. trans-1-iodo-4-ethylcyclohexane and methoxide ion
b. cis-1-chloro-3-methylcyclobutane and ethoxide ion
Question:Draw the elimination products for each of the following E2 reactions; if the products can exist as stereoisomers, indicate which stereoisomers are obtained.
a. (2S,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
b.(2S,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
c.(2R,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
d.(2R,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
e.3-chloro-3-ethyl-2,2-dimethylpentane + high concentration of CH3CH2O-
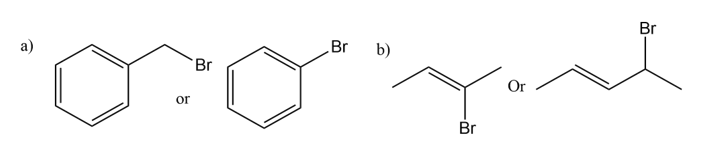
For the pairs of compounds in Problem 48, which is more reactive in an SN2 reaction?
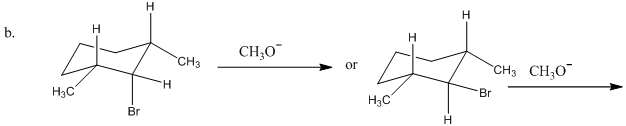
Which reactant in each of the following pairs undergoes an elimination reaction more rapidly? Explain your choice.

What do you think about this solution?
We value your feedback to improve our textbook solutions.