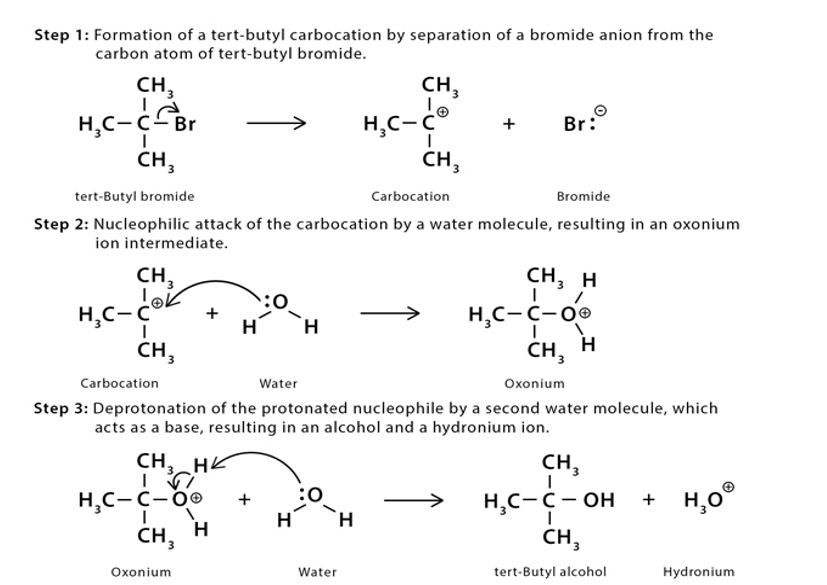
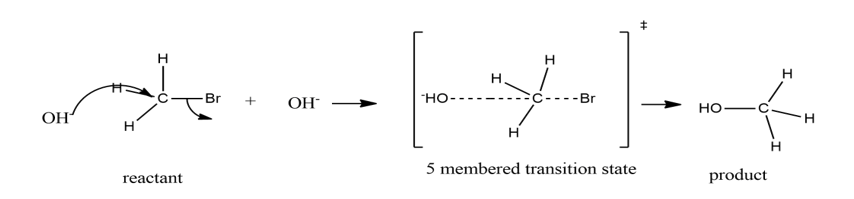
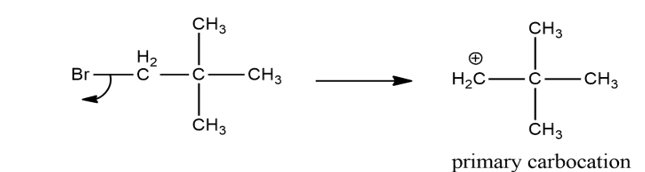
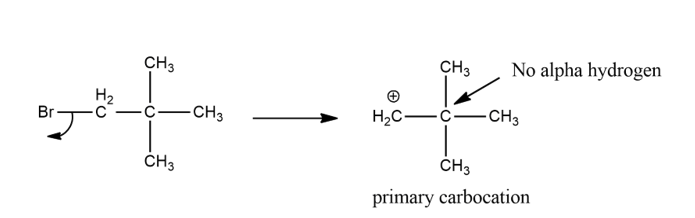
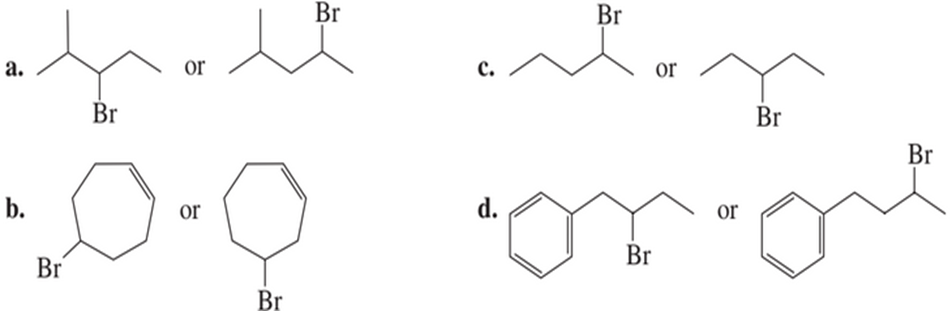
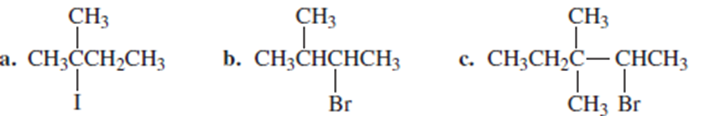
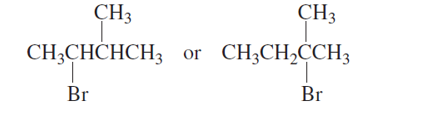Chapter 9: Q43P (page 433)
a. Explain why 1-bromo-2,2-dimethylpropane has difficulty undergoing both SN2 and SN1 reactions.
b. Can it undergo E2 and E1 reactions?
Short Answer
a) 1-bromo-2,2-dimethylpropane doesn’t undergo elimination reaction as C-Br and C-H bonds are not in anti-periplanar position.
b) It can’t undergo E2 and E1 reaction as it doesn’t have an 𝜶 H to undergo elimination with C-Br bond.