Chapter 1: Q59P (page 2)
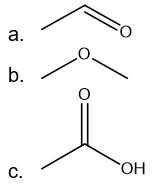
Draw the skeletal structure for each of the compounds
1. CH3CHO
2. CH3OCH3
3. CH3COOH
Short Answer
Learning Materials
Features
Discover
Chapter 1: Q59P (page 2)
Draw the skeletal structure for each of the compounds
1. CH3CHO
2. CH3OCH3
3. CH3COOH

All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the partial negative charge on the fluorine atom in a C-F bond. The bond length is 1.39 1.39A* , and the bond dipole moment is 1.06D. The charge on an electron is 4.80⨯10-10esu.
Describe the orbital used in bonding and the bond angles in the following compounds:
a. CH3O-
b. CO2
c. H2CO
d. N2
e. BF3
Which of the following molecules would you expect to have a dipole moment of zero? To answer parts g and h, you may need to review the Problem Solving Strategy on p. 39.

Draw the ground-state electronic configuration for each of the following:
a. Mg
b.Ca2+
c. Ar
d. Mg2+
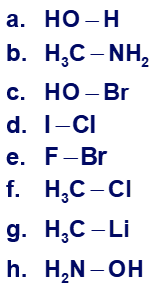
Use the symbol and to show the direction of polarity of the indicated bonds in each of the following compounds:
What do you think about this solution?
We value your feedback to improve our textbook solutions.