Chapter 15: Q15-2P (page 690)
Name the following:
Short Answer
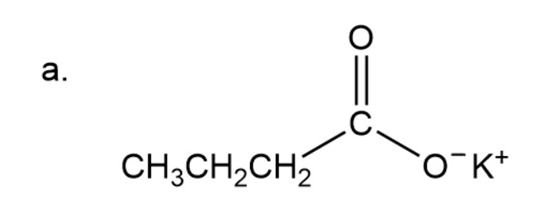
(a)Potassium butanoate
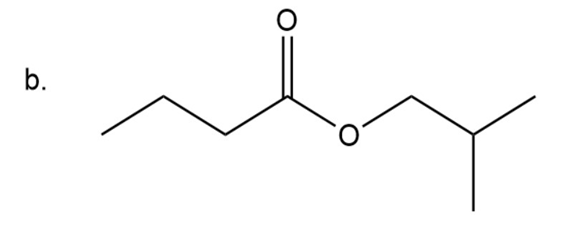
(b)Isobutyl butanoate
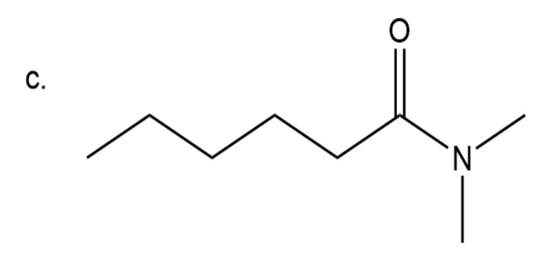
(c)N,N-dimethylhexanamide
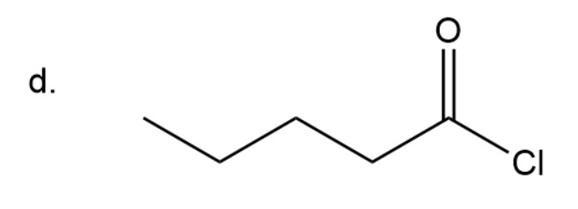
(d) Pentanoyl chloride
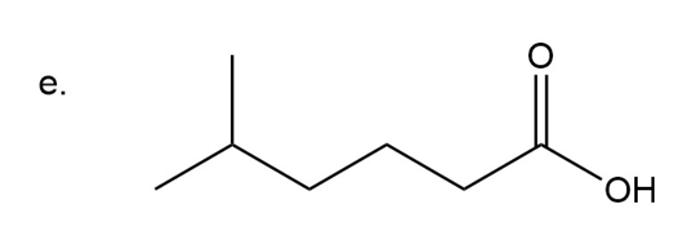
(e) 5-methylhexanoic acid
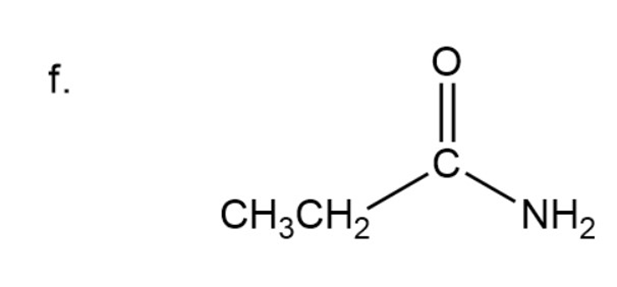
(f) Propanamide
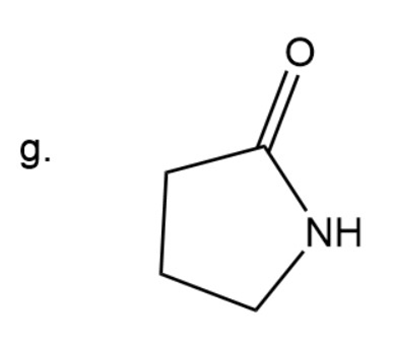
(g) 2-Azacyclopentanone
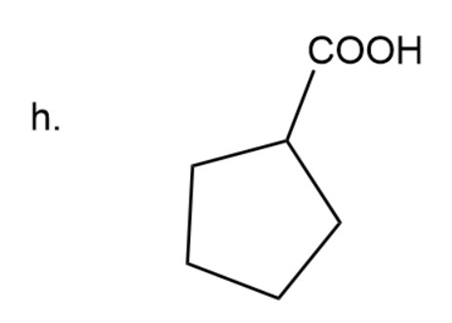
(h) Cyclopentanecarboxylic acid
(i) 5-Methyl-2-oxacyclohexanone
Step by step solution
Carboxylic acid derivatives
Carbonyl groups containing compounds other than an aldehyde or ketone are called carboxylic acid derivatives.
Naming of salt of carboxylic acid
Here cation is named first then followed by name of acid and at the end ‘ate’ is used instead of ‘ic acid’.
In the given compound potassium is a cation and the acid has four carbon so will be butyl.

Potassium butanoate
The IUPAC name of compound is potassium butanoate.
The common name of the compound is potassium butyrate.
Naming of ester
Esters have two alkyl groups: one is present attached to carbonyl group and other is attached to carboxyl oxygen. The group attached to carboxyl oxygen is written first then followed by parent chain naming and at last ‘ate’ is used instead of ‘ic acid’.
Here on carboxyl oxygen four carbon chain with iso-configuration is present so it is isobutyl group and the chain including carbonyl group is also having four carbon it is butyl group.
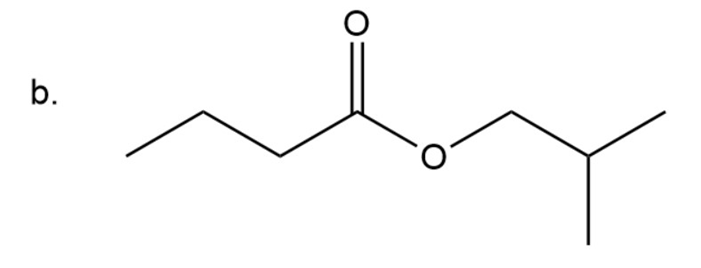
Isobutyl butanoate
The IUPAC name of compound is isobutyl butanoate.
Common name of compound is isobutyl butyrate.
Step 4: Naming of amide
In amide carbonyl group is present adjacent to instead of OH group. In amide instead of ‘oic acid’, ‘amide’ is used at the end.
Here two methyl groups are attached to amide nitrogen and six carbon chain is
present.
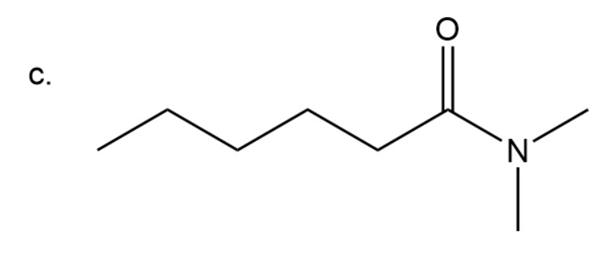
N,N-dimethylhexanamide
The IUPAC name of compound isN,N-dimethylhexanamide.
Common name of compound isN,N-dimethylhexanamide.
Naming of Acyl chlorides
In amide carbonyl group is attached to chlorine instead of OH group. In acyl chloride instead of ‘ic acid’, ‘yl chloride’ is used at the end. In case of cyclic acid carboxylic acid is replaced by carbonyl chloride.
Here chain has five carbon.
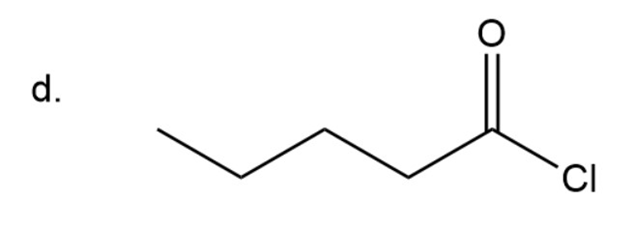
Pentanoyl chloride
The IUPAC name of compound isPentanoyl chloride.
Common name of compound is valeryl chloride.
Step 6: Naming of carboxylic acid
In carboxylic acid carbonyl group is attached to OH group. Here the name of acid is written according to carbon present in chain and at the end ‘oic acid’ is used.
Chain has six carbon with methyl group substitution at carbon number five.In common name are used for numbering carbon adjacent to carbonyl group as is used for carbon adjacent to carbonyl group, for carbon adjacent to and so on.
Here methyl group is present four carbon away from carbonyl group so is used in naming
.
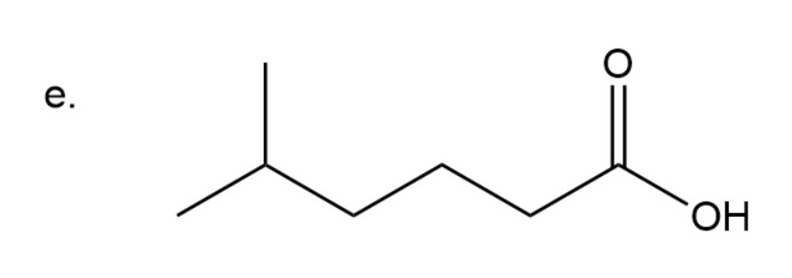
5-methylhexanoic acid
The IUPAC name of compound is5-methylhexanoic acid.
The common name of the compound is 8-methylcaproic acid.
Naming of amide
Here carbonyl group is present adjacent to group instead of OH group so it is a amide. In amide instead of ‘oic acid’, ‘amide’ is used at the end.
Here three carbon are present in chain.
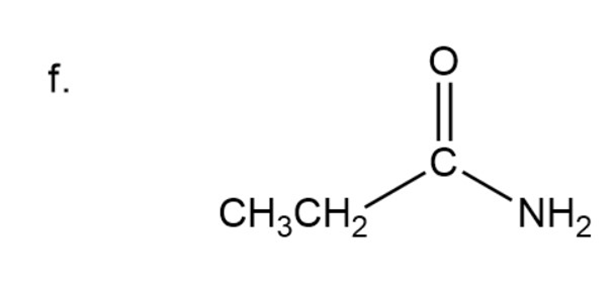
Propanamide
The IUPAC name of compound isPropanamide.
Common name of compound is Propionamide.
Naming of ketone
Here carbonyl group is present adjacent to two alkyl group. In ketone ‘one’ is used at the end. The prefix aza- is used to give names to compounds where a carbon is replaced by a nitrogen atom.
Here ring is five membered.
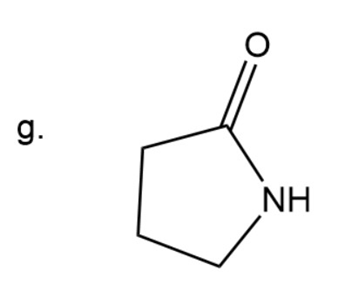
2-Azacyclopentanone
The IUPAC name of compound is2-Azacyclopentanone.
Common name of compound is- gama butyrolactam.
Naming of cyclic carboxylic acid
In carboxylic acid carbonyl group is attached to OH group. Here the name of acid is written according to carbon present in chain and at the end ‘carboxylic acid’ is used.
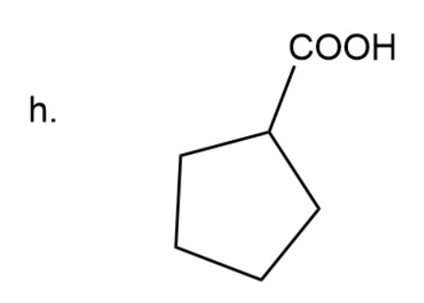
Cyclopentanecarboxylic acid
The IUPAC name of compound iscyclopentanecarboxylic acid.
Common name of compound is cyclopentanecarboxylic acid.
Naming of ketone
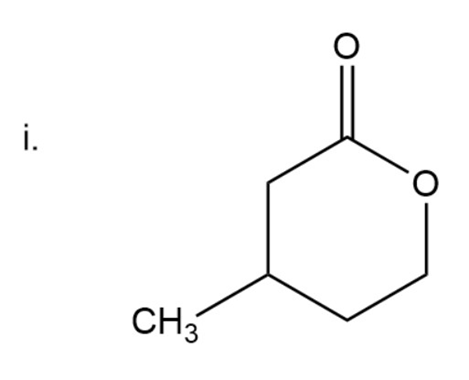
Here carbonyl group is present next to two alkyl groups. In ketone ‘one’ is used at the end of the name. Oxa is used in compounds where a carbon atom is replaced by oxygen.
Here the ring is six-membered.
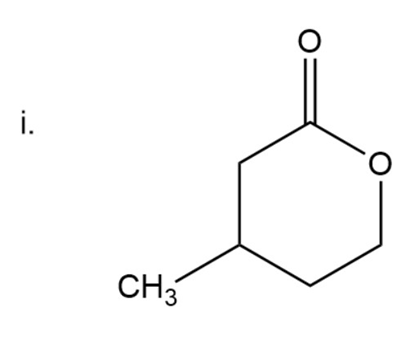
5-Methyl-2-oxacyclohexanone
The IUPAC name of compound is 5-Methyl-2-oxacyclohexanone.
Common name of compound is beta--Methyl and gama-valerolactone.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!