Chapter 18: Q98P (page 921)
a. Explain why the following reaction leads to the products shown:
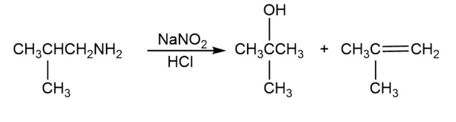
b. What product is obtained from the following reaction?
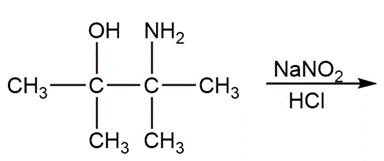
Short Answer
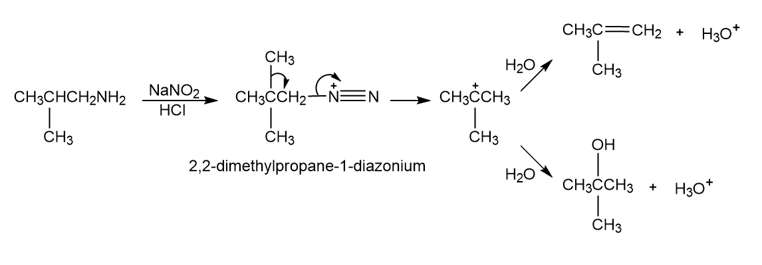
a.
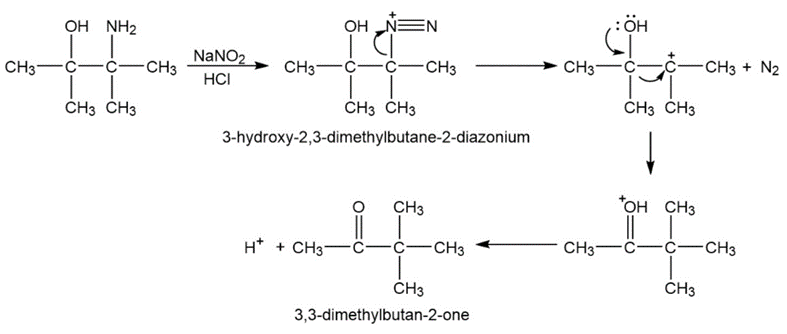
b.
Learning Materials
Features
Discover
Chapter 18: Q98P (page 921)
a. Explain why the following reaction leads to the products shown:

b. What product is obtained from the following reaction?

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Describe how mescaline can be synthesized from benzene. The structure of mescaline is given on page 869.
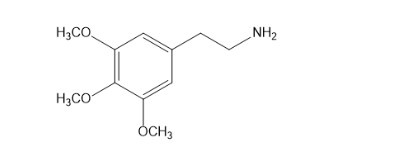
What is the major product of a Friedel-crafts alkylation using the following alkyl chlorides?
a. CH3CH2Cl
b. CH3CH3CH2Cl
c. CH3CH2CH(Cl)CH3
d. (CH3)3CCl
e. (CH2)2CHCH2Cl
f. CH2CHCH2Cl
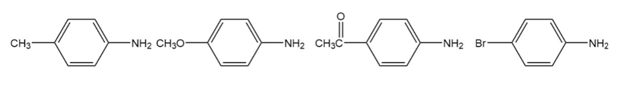
Rank the following substituted anilines from most basic to least basic:
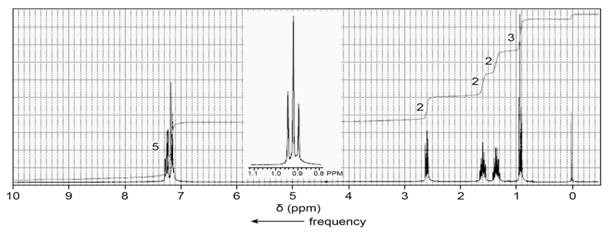
Benzene underwent a Friedel–Crafts acylation followed by a Wolff–Kishner reduction. The product gave the following 1H NMR spectrum. What acylchloride was used in the Friedel–Crafts acylation?
a. Rank the following compounds from greatest tendency to least tendency to undergo nucleophilic aromatic substitution:
chlorobenzene 1-chloro-2,4-dinitrobenzene p-chloronitrobenzene
b. Rank the same compounds from greatest tendency to least tendency to undergo electrophilic aromatic substitution.
What do you think about this solution?
We value your feedback to improve our textbook solutions.