Question: The pKa values of a few ortho-, meta-, and para-substituted benzoic acids are shown below:
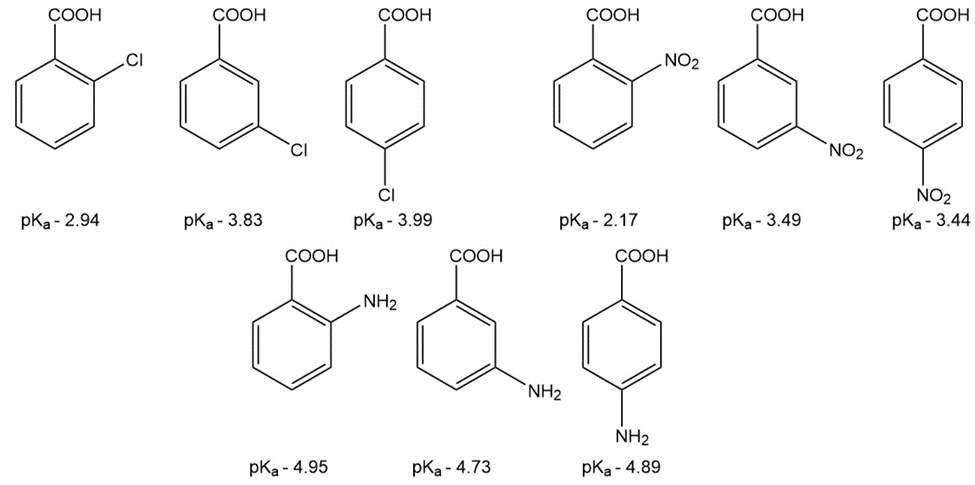
The relative pKa values depend on the substituent. For chloro-substituted benzoic acids, the ortho isomer is the most acidic and the para isomer is the least
acidic; for nitro-substituted benzoic acids, the ortho isomer is the most acidic and the meta isomer is the least acidic; and for amino-substituted benzoic acids, the meta isomer is the most acidic and the ortho isomer is the least acidic. Explain these relative acidities.
a. Cl: ortho > meta > para
b. NO2: ortho > para > meta
c. NH2: meta > para > ortho


















