Chapter 18: Q37P (page 906)
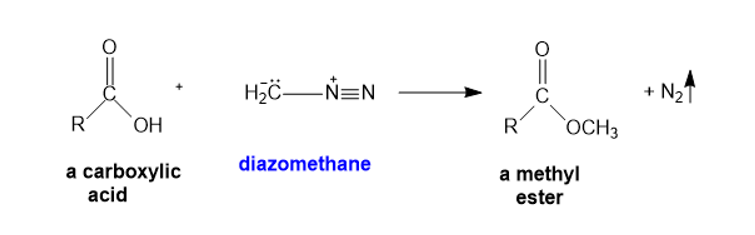
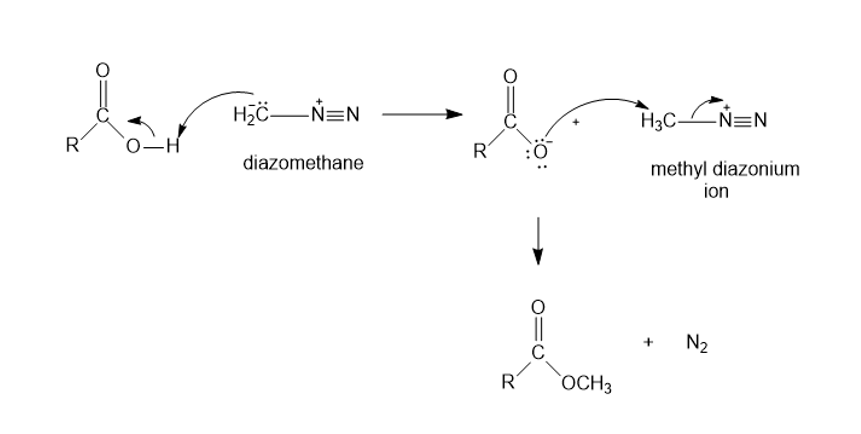
Diazomethane can be used to convert a carboxylic acid to a methyl ester. Propose a mechanism for this reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q37P (page 906)
Diazomethane can be used to convert a carboxylic acid to a methyl ester. Propose a mechanism for this reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
a. Explain why the following reaction leads to the products shown:
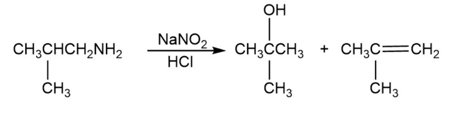
b. What product is obtained from the following reaction?
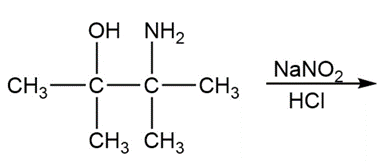
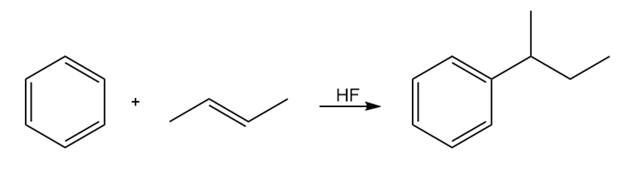
Friedel–Crafts alkylations can be carried out with carbocations formed from reactions other than the reaction of an alkyl halide with AlCl3. Propose amechanism for the following reaction:
Describe two synthetic routes for the preparation of p-methoxyaniline from benzene.
Show how the following compounds can be synthesized from benzene:
a. m-chlorobenzenesulfonic acid
b. m-chloroethylbenzene
c. m-chlorobenzonitrile
d. 1-phenylpentane
e. m-bromobenzoic acid
f. m-hydroxybenzoic acid
g. p-cresol
h. benzyl alcohol
i. benzylamine
What do you think about this solution?
We value your feedback to improve our textbook solutions.