Chapter 18: Q83P (page 919)
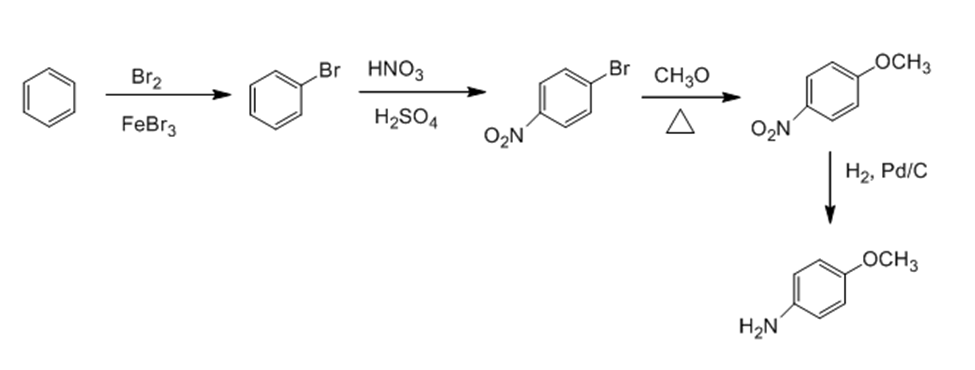
Describe two synthetic routes for the preparation of p-methoxyaniline from benzene.
Short Answer

Learning Materials
Features
Discover
Chapter 18: Q83P (page 919)
Describe two synthetic routes for the preparation of p-methoxyaniline from benzene.


All the tools & learning materials you need for study success - in one app.
Get started for free
Why isn’t FeBr3 used as a catalyst in the first step of the synthesis of 1,3,5-tribromobenzene?
A student had prepared three ethyl-substituted benzaldehydes, but neglected to label them. The student at the next bench said they could be identified by brominating a sample of each and determining how manybromo-substituted products are formed. Is the student’s advice sound?
In the mechanism for electrophilic aromatic substitution with a diazonium ion as the electrophile, why does nucleophilic attack occur on the terminal nitrogen of the diazonium ion rather than on the nitrogen that hasthe formal positive charge?
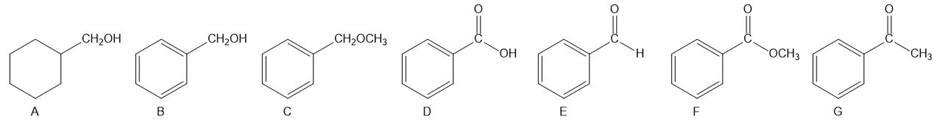
How can you distinguish the following compounds using:
a. their infrared spectra?
b. their 1 H NMR spectra?
Show how the following compounds can be synthesized from benzene:
a. m-chlorobenzenesulfonic acid
b. m-chloroethylbenzene
c. m-chlorobenzonitrile
d. 1-phenylpentane
e. m-bromobenzoic acid
f. m-hydroxybenzoic acid
g. p-cresol
h. benzyl alcohol
i. benzylamine
What do you think about this solution?
We value your feedback to improve our textbook solutions.