Chapter 18: Q35P (page 906)
Which amide bond is hydrolyzed in the first step of the conversion of temozolomide to methyldiazonium?
Short Answer

Learning Materials
Features
Discover
Chapter 18: Q35P (page 906)
Which amide bond is hydrolyzed in the first step of the conversion of temozolomide to methyldiazonium?

All the tools & learning materials you need for study success - in one app.
Get started for free
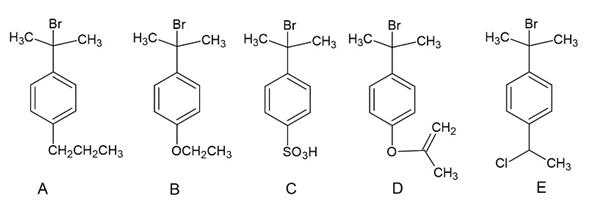
The following tertiary alkyl bromides undergo anreaction in aqueous acetone to form the corresponding tertiary alcohols. Rank the alkyl bromides from most reactive to least reactive.
Tyramine is an alkaloid found in mistletoe and ripe cheese. Dopamine is a neurotransmitter involved in the regulation of the central nervous system.
 Tyramine Dopamine
Tyramine Dopamine
a. How can tyramine be prepared from \({\bf{\beta }}\)-phenylethylamine?
b. How can dopamine be prepared from tyramine?
c. Give two ways to prepare \({\bf{\beta }}\) -phenylethylamine from \({\bf{\beta }}\)-phenylethyl chloride.
d. How can \({\bf{\beta }}\)-phenylethylamine be prepared from benzyl chloride?
e. How can \({\bf{\beta }}\)- phenylethylamine be prepared from benzaldehyde?
Using resonance contributors for the carbocation intermediate, explain why a phenyl group is an ortho–para director.
Describe two synthetic routes for the preparation of p-methoxyaniline from benzene.
What are the products of the following reactions?

What do you think about this solution?
We value your feedback to improve our textbook solutions.